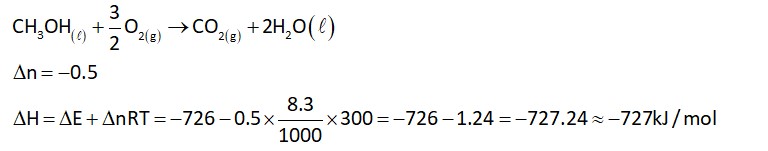Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
6 months agoContributor-Level 10
Here, total meq of acetic acid = 50 * 0.1 = 5
And total meq of NaOH = 25 * 0.1 = 2.5
After neutralization process
Meq of left acetic acid = 2.5
And meq of formed CH3COONa = 2.5
New answer posted
6 months agoContributor-Level 10
Baking soda = NaHCO3
Washing soda = Na2CO3. 10H2O
Caustic soda = NaOH
New answer posted
6 months agoContributor-Level 10
HOCl produce in the stratospheric cloud, by the hydrolysis reaction of ClONO2.
New answer posted
6 months agoContributor-Level 10
Due to H- bond in water, it has high melting point and melting point of other hydrides of the group are depending upon the molecular weight.
New answer posted
6 months agoContributor-Level 10
Due to high crystallity Be has the highest M.P.
Be = 1560 K
Mg = 925 K
Ca = 1120 K
Sr = 1062 K
New answer posted
6 months agoContributor-Level 10
In boron family lower O.S (+1) is more stabilize down the group due to inert pair effect.
New answer posted
6 months agoContributor-Level 10
Wt of liquid = 135 – 40 = 95 gm
Volume of liquid =
Hence volume of vessel = 100 ml = 0.1 lit from ideal gas equation,
New answer posted
6 months agoContributor-Level 9
For expansion in vacuum, workdone, w = 0
For isothermal process,
According to first law of thermodynamics,
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers