Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
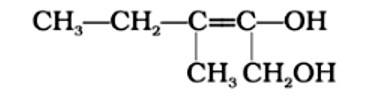
3-Methylpent-2-ene-1,2-diol
New answer posted
11 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
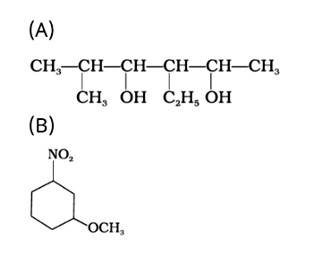
The IUPAC name of the given compound is:
(A) 3-Ethyl-5-methylhexane-2,4-diol
(B) 1-Methoxy-3-nitrocyclohexane
New answer posted
11 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
The structure of glycerol is:
CH2—CH—CH2
| | |
OH OH OH
The IUPAC of glycerol is Propane-1,2,3-triol.
New answer posted
11 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
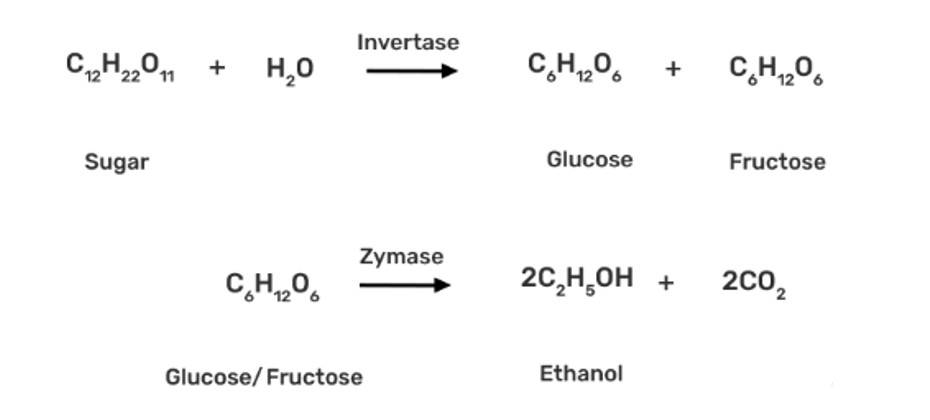
Commercially, ethanol (C2H5OH) is made by fermenting carbohydrates, which is the earliest process. In the presence of an enzyme called invertase, sugar in molasses, sugarcane, or fruits like grapes is transformed to glucose and fructose (both of which have the formula C6H12O6).
In the presence of another enzyme, zymase, present in yeast, glucose and fructose are fermented.
Grapes are used to make wine because they contain sugars and yeast. The amount of sugar in grapes increases as they ripen, and yeast forms on the outer peel. When grapes are crushed, sugar and enzymes c
New answer posted
11 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
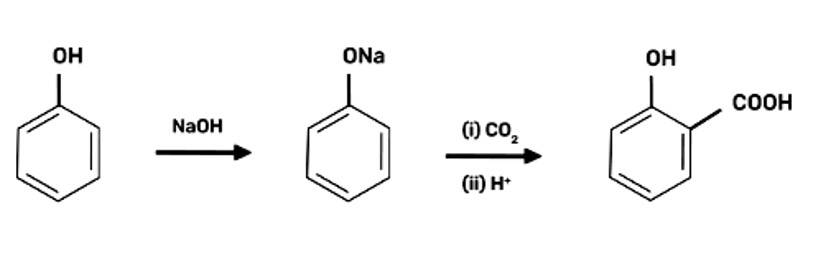

2-Hydroxybenzoic acid
&nb
New answer posted
11 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
(a) Cumene is the beginning element for the industrial production of phenol.
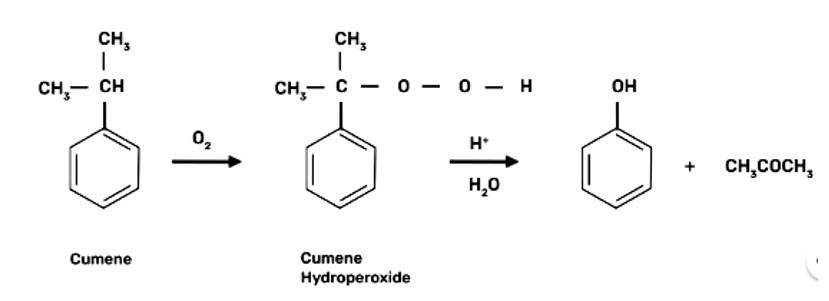
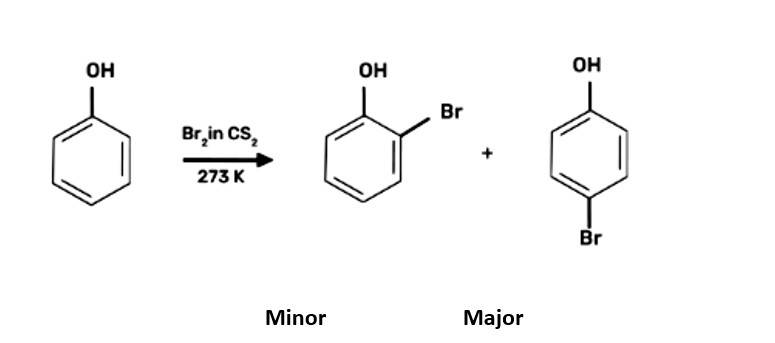
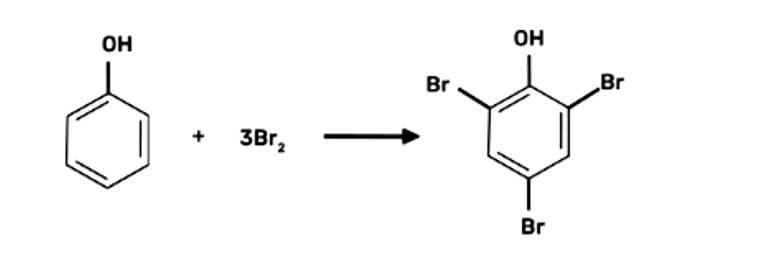
When bromine water is used to treat phenol. As a whitish precipitate, 2,4,6-tribromophenol is formed
New answer posted
11 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
In case of anisole, methylphenyl oxonium ion, 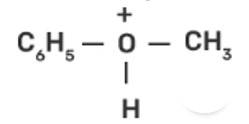
New answer posted
11 months agoContributor-Level 10
This is a Fill in the blanks Type Question as classified in NCERT Exemplar
Correct option: C
A catalyst allows a chemical reaction to occur at a faster rate or under different conditions than it would otherwise. As a result, a catalyst affects the reaction's enthalpy change, or heat. As a result, in the presence of the reaction, the enthalpy does not vary, i.e., it remains constant, and no heat is produced or absorbed.
New answer posted
11 months agoContributor-Level 10
This is a Fill in the blanks Type Question as classified in NCERT Exemplar
Correct option: B
The Arrhenius equation can be used to calculate the activation energy of a chemical process. At two temperatures, this determines the rate constants.
2.303log = =
New answer posted
11 months agoContributor-Level 10
This is a Fill in the blanks Type Question as classified in NCERT Exemplar
Option C
Gibbs energy of reaction - Gibbs free energy is a single-valued combination of entropy and enthalpy. The direction of a chemical reaction at constant temperature and pressure is predicted by Gibbs free energy.
Enthalpy of reaction – When one mole of matter is converted by a chemical reaction under specified conditions, the enthalpy of reaction is the change that occurs in the system.
Activation energy of reaction – The lowest amount of energy necessary to activate molecules to a state where they can perform physical and chemical transformations is known
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers