Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i)Conductivity k is defined as k =
(ii)Molar conductivity is given by Λm =
(iii)Degree of dissociation is given by ? =
(iv)Charge is the product of current and time = I
Hence, the answer is:
(i)-d; (ii)-c; (iii)-b; (iv)-a
New answer posted
11 months agoContributor-Level 10
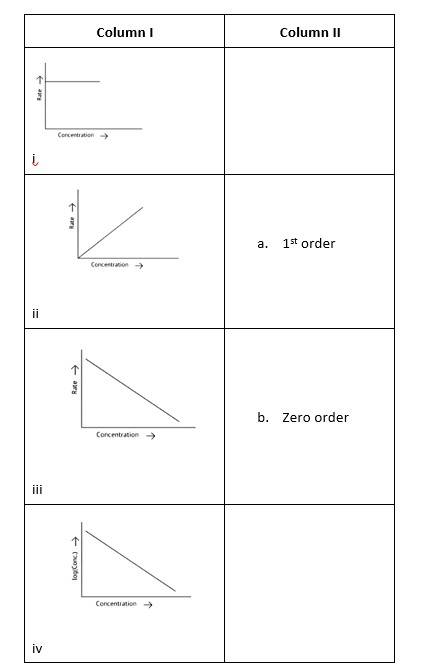
This is a Matching Type Question as classified in NCERT Exemplar
(i)- (a) ; (ii)- (b) ; (iii)- (b); (iv)- (a)
A zero-order reaction is one in which the reactant concentrations do not change over time and the rate of concentration remains constant.
A first-order reaction is one in which the rate of the reaction is linearly proportional to the concentration of only one ingredient. In other terms, a first-order reaction is a chemical reaction whose rate is determined by changes in only one of the reactants' concentration.
New answer posted
11 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i) The cell reaction of a lead storage battery is as follows;
Pb + PbO2 + 2H2SO4→2H2SO4 + 2H2O
At cathode:PbO2 (s)+SO42− (aq)+2e−→2PbSO4 (s) + 2H2O (l)
At anode:Pb (s)+SO42− (aq)→PbSO4 (s)+2e−
Therefore Pb is the anode and PbO2 is cathode
(ii)Mercury cell doesn't contain ions so it gives steady potential.
(iii)Fuel cells have maximum efficiency as they produce energy due to the combustion reaction of fuel.
(iv)Rusting is prevented by galvanization.
Hence, the answer is:
(i)-d ; (ii)-c ; (iii)-a ; (iv)-b
New answer posted
11 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i)On dilution, the number of ions per unit volume increases so the molar conductivity increases with dilution.
(ii) E cell is an intensive property as it is not dependent on the amount or mass of the substance.
(iii) k is conductivity or be precise specific conductivity which depends on the number of ions
(iv) ΔrGcell is an extensive property as it depends on the amount of substance or number of particles in the solution.
Hence, the answer is:
(i)-d ; (ii)a ; (iii)-b ; (iv)-c
New answer posted
11 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Ans: (i)The unit of Λm is Scm2mol−1
(ii) The unit of Ecell is m−1
(iii) The unit of k is Scm−1
(iv)The unit of G∗ is m−1
Hence, the answer is:
(i)-c ; (ii)-d ; (iii)-a ; (iv)-b
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: A and D
The pace of a reaction that is directly proportional to the concentration of the reacting substance is known as a first order reaction.

Y=mn
k = log
t = log
x= a-
x=
t = log
log2
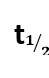
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: A and D
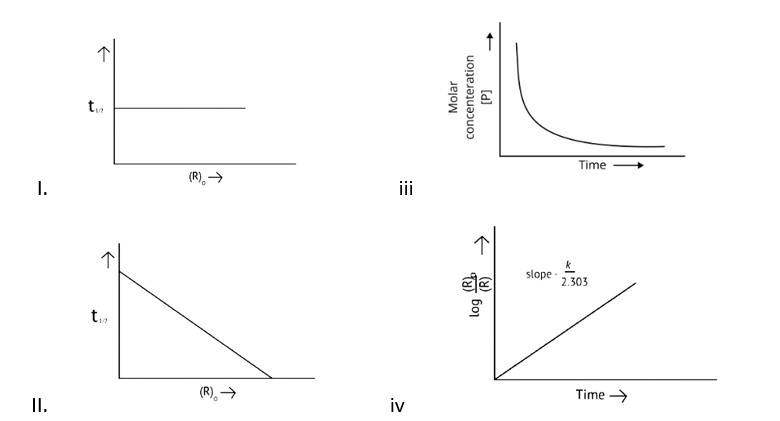
A Zero order reaction = [R] = ( - k)t + [R]0
y = (m * ) + c
x = t (time)
y = [R]concentration
Slope (m) = - k
Intercept (c) = [R]0
= - k
= - kto
Rate ∝ t0
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: Option (ii, iii)
In the given representation of the cell, the left side represents the oxidation half cell and the right side of the cell represents the reduction half cell
The electrode at which oxidation occurs is called anode and oxidation of magnesium occurs at anode so magnesium is the anode.
Similarly, the electrode at which the reduction occurs is called the cathode, and the reduction of copper occurs at the cathode.
The cell reaction is as follows
Mg + Cu2+→Mg2++ Cu
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: Option (i, iii)
The conductivity in an ionic solution is due to the ions in the solution. It depends on the following factors;
(a)Temperature: On an increase in temperature, the molar conductivity of ionic solutions increases.
(b)Concentration of Electrolytes: An increase in the concentration of electrolytes decreases the molar conductivity as the number of ions per unit volume decreases.
Therefore, options (i, iii) are correct
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options: B, C and D
Catalysts reduce the activation energy of a process and give an alternate path by reducing or raising the activation energy between reactants and products, altering the reaction's enthalpy change. As a result, B, C, and D statements are the proper responses
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers