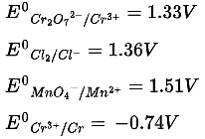Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct Option: (iii)
Ans: Given: One mole of aluminium
Calculate the number of electrons required to convert Al2O3 to Al
It is the charge required to obtain one mole of aluminium from Al2O3
The dissociation of aluminium oxide is as follows;
Al2O3→2Al3++3O2−
Al3++3e−→Al
Therefore 3Fcharge is required to obtain one mole of aluminum from Al2O3.
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (i)
According to the given table, ![]() has the most negative value among the given species. So
has the most negative value among the given species. So
Cr3+ is the most stable oxidized species.
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Correct options : A, C and D.
Rate law = Experimental rate of the reaction
Suppose aA + bB→cC + dD
Then rate = k [A] α [B] β
Here instead of using complex reaction we use elementary reaction which gets solved in one step
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iv)
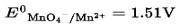
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iii)
According to the electrochemical series, the lower the reduction potential higher is the reducing power.
Therefore, the order of reducing power is Mn2+ < Cl < Cr3+ < Cr
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (iii)
According to the electrochemical series higher the positive value of standard reduction potential of metal ion, the higher is the oxidizing power
Therefore, MnO4− is the strongest oxidizing agent.
New answer posted
11 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Incorrect order results from a balanced chemical equation. KClO3 + 6FeSO4 + 3H2SO4→KCl + 3H2O + 3Fe2 (SO4)3
This reaction is complex and occurs in several steps. The order is determined by the slowest in the reactions
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct Option: (ii)
According to the electrochemical series and standard reduction potential of metal, the higher the negative value of standard reduction potential stronger will be the reducing agent
In the given options, the standard reduction potential of chromium has the highest value so it is the strongest reducing agent.
New answer posted
11 months agoContributor-Level 10
This is a Short Answer Type Question as classified in NCERT Exemplar
Simple reactions occur in a single stage, whereas complicated reactions occur in multiple steps. Each phase in a multi-step process has its own molecularity, which is not added together. There is no molecularity in complex reactions. The sequence of a complex reaction is decided by the slowest step. As a result, it can be used for both simple and complex processes
New question posted
11 months agoTaking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers