Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
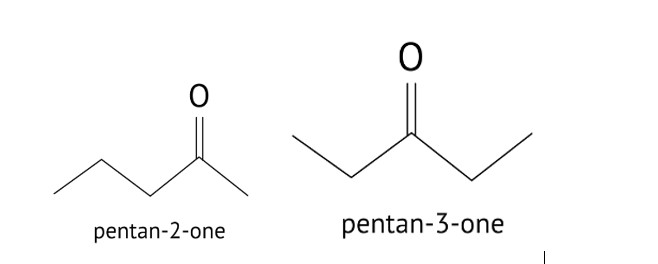
Butanol is a four-carbon aldehyde, with the aldehyde group on the fourth carbon. It has a functional group and a carbonyl group that interacts dipole-dipole. Because butanol possesses a polar O-H bond, it exhibits intermolecular H bonding, which is not feasible in butanal due to the lack of a polar bond. The boiling point of butanol is higher than that of butan-1-ol.
New answer posted
9 months agoContributor-Level 10
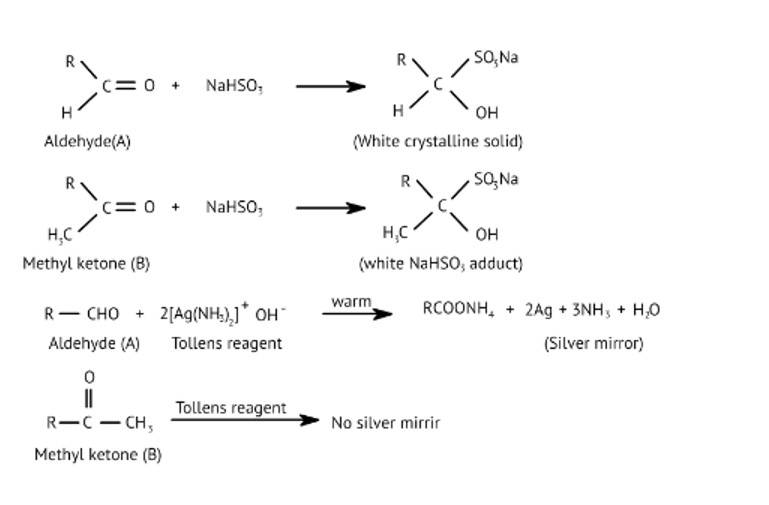
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: Liquid 'A' reduces ammoniacal silver nitrate and 'B' is a ketone which forms white crystalline solid on treatment with hydrogen sulphite
New answer posted
9 months agoContributor-Level 10
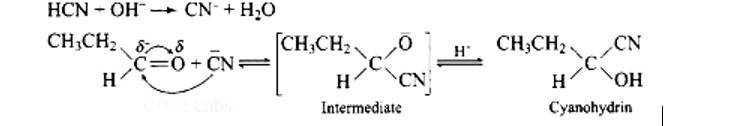
This is a Long Answer Type Questions as classified in NCERT Exemplar
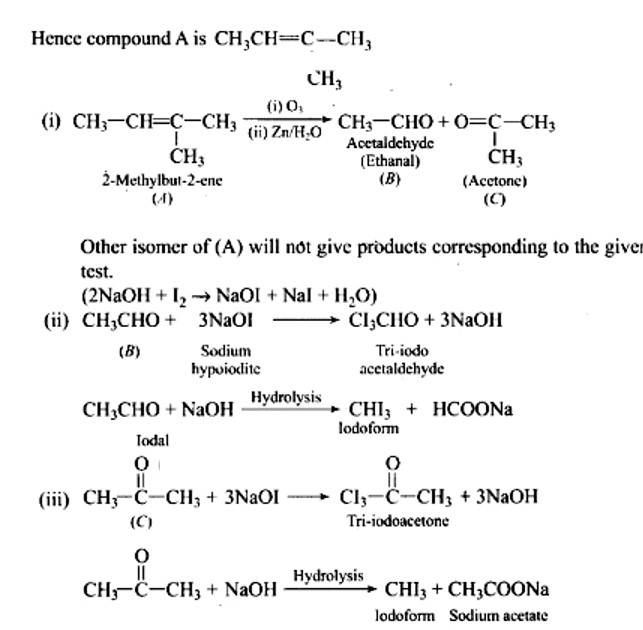
Ans: Functional isomers

Compound I will react faster with HCN due to less steric hindrance and greater positive charge on carbon atom of carbonyl group. Two methyl groups increase electron density on carbonyl carbon in compounds II hence the rate of nucleophilic attack is less.
Mechanism of the reaction:
New answer posted
9 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans: The aromatic compound 'A' does not give Tollen's reagent test, it is not an aromatic aldehyde. As it responds to an iodoform test called methyl ketone.
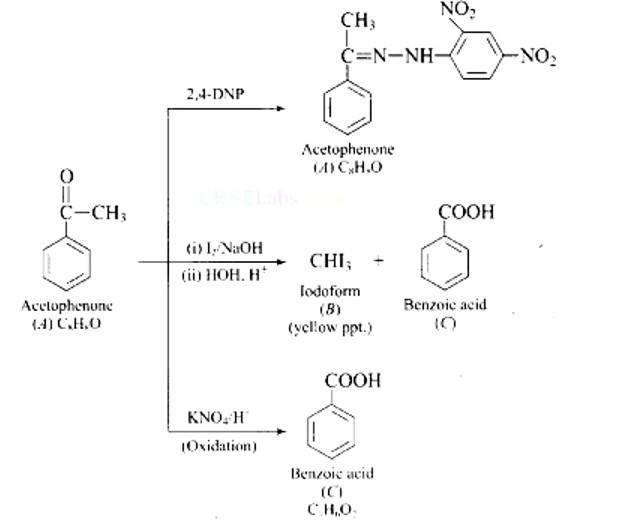
New answer posted
9 months agoContributor-Level 10
This is a Long Answer Type Questions as classified in NCERT Exemplar
Ans:
Fehling's test is positive when compound 'B' is used. It gives an iodoform test and proves that it is an aldehyde. Because it fails Fehling's test, compound 'C' is a ketone.
New answer posted
9 months agoContributor-Level 10
According to the de Broglie equation, the particles like electrons have wave-like properties. The quantum mechanical model foundation is laid by this concept and it also explains the stability of electron orbits using wave behavior.
New answer posted
9 months agoContributor-Level 10
The hydrogen atom can be accurately explained by Bohr's model. However, it does not account for shielding effects, electron-electron interactions, and the wave nature of electrons for multi-electron atoms.
New answer posted
9 months agoContributor-Level 10
Quantum numbers describe the unique position and energy of an electron in an atom. These are a set of four numbers and are important for predicting chemical behavior and understanding the distribution of electrons in orbitals.
New answer posted
9 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Ans: Correct option: D
[Fe (CN)6]3- ion shows magnetic moment corresponding to one unpaired electron. (CN) is a ligand with a strong field that couples electrons, resulting in hybridisation. d2sp3 and it has one unpaired electron, it has the magnetic moment of one paired electron.
μ= = 1 (1 + 2)
= 3 = 1.73BM
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers