Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
The structures of cis- and trans-isomer of hex-2-ene are:
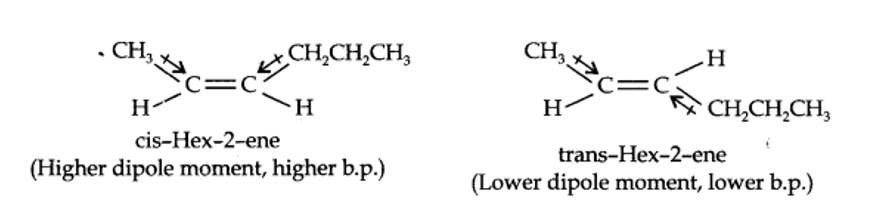
The boiling point of a molecule depends upon dipole-dipole interactions. Since cis-isomer has higher dipole moment, therefore, it has higher boiling point.
New answer posted
11 months agoContributor-Level 10
3.40. Within a period, the oxidising character increases from left to right. Therefore, among F, O and N, oxidising power decreases in the order: F > O > N. However, within a group, oxidising power decreases from top to bottom. Thus, F is a stronger oxidising agent than Cl. Further because O is more electronegative than Cl, therefore, O is a stronger oxidising agent than Cl. Thus, overall decreasing order of oxidising power is: F > O > Cl > N, i.e., option (b) is correct.
New answer posted
11 months agoContributor-Level 10
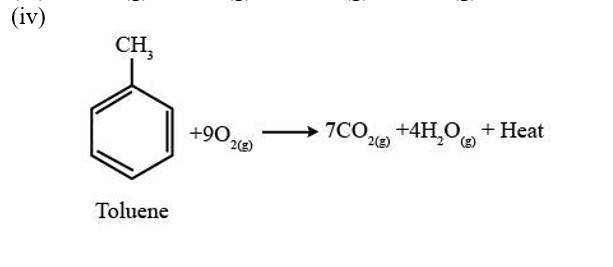
A combustion reaction is a reaction in which a substance reacts with oxygen gas, there is a formation of carbon dioxide, water with the evolution of light and heat.
(i) 2C4H10 (g) +13 O2 (g)?8CO2 (g)+10H2O (g) + Heat
(ii) 2C5H10 (g) +15 O2 (g)?10CO2 (g)+10H2O (g) + Heat
(iii) 2C6H10 (g) +17 O2 (g)?12CO2 (g)+10H2O (g) + Heat
New answer posted
11 months agoContributor-Level 10
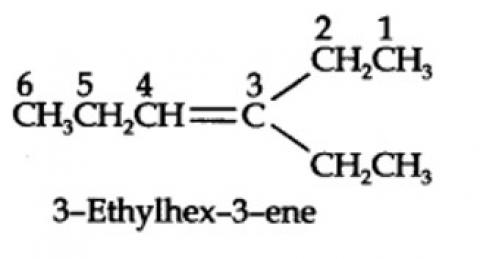
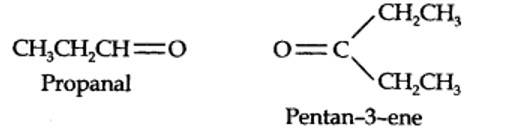
The ozonolysis of 4-Ethylhex-3-ene gives propanal and pentan-3-one.
The structural formula of the alkene (4-Ethylhex-3-ene) is as shown.
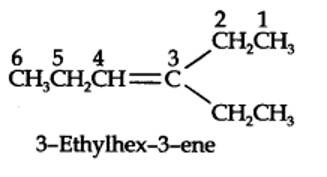
New answer posted
11 months agoContributor-Level 10
1.44 Ge is group 14 element and In is group 13 element. Hence an electron deficient hole is created and therefore, it is p–type.
2. B is group 13 elements and Si is group 14 elements, there will be a free electron. Hence, it is n-type
New answer posted
11 months agoContributor-Level 10
3.39. In a period, the non-metallic character increases from left to right. Thus, among B, C, N and F, non-metallic character decreases in the order: F > N > C > B. However, within a group, non-metallic character decreases from top to bottom. Thus, C is more non-metallic than Si. Therefore, the correct sequence of decreasing non-metallic character is: F > N > C > B > Si, i.e., option (c) is correct.
New answer posted
11 months agoContributor-Level 10
(i) An aldehyde with molar mass of 44 u is ethanal, CH3CH=0
(ii) Write two moles of ethanal side by side with their oxygen atoms pointing towards each other.
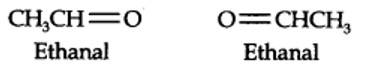
New answer posted
11 months agoContributor-Level 10
3.38. In a period, metallic character decreases as we move from left to right. Therefore, metallic character of K, Mg and Al decreases in the order: K > Mg > Al. However, within a group, the metallic character, increases from top to bottom. Thus, Al is more metallic than B. Therefore, the correct sequence of decreasing metallic character is: K > Mg > Al > B, i.e., option (d) is correct
New answer posted
11 months agoContributor-Level 10
1.43 There is one octahedral hole for each atom in hexagonal close packed arrangement. If the number of oxide ions (O2−) per unit cell is 1, then the number of

New answer posted
11 months agoContributor-Level 10
Step 1. Write the structure of the products side by side with their oxygen atoms pointing towards each other.
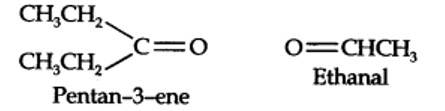
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers
