Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
(i) Both metallic and ionic crystals have strong forces of attraction between their atoms or ions. In metallic crystals there is a strong metallic bond present between electrons and positively charged ions. In ionic crystals there are strong ionic bonds between anions and cations. Both of them conduct electricity,
but metallic crystals conduct electricity in all three states of matter while ionic crystals conduct electricity only in molten state.
(ii) The ionic solids are hard and brittle because they have strong electrostatic forces of attraction between the anions and cations .As a result the anions and cations are tightly hel
New answer posted
11 months agoContributor-Level 10
(d) Chlorofluorocarbons (CFCs) are man-made industrial chemicals used in air conditioning etc.
New answer posted
11 months agoContributor-Level 10
1.32 Number of corner atoms per unit cell
= 8 corners atoms *18 atom per unit cell
=8 * 18 = 1 atom
Number of face face centred atoms per unit cell
= 6 face centred atoms * 12 atom per unit cell
= 6 * 12 = 3 atoms
∴ Total number of atoms (lattice point) = 1 + 3 = 4
(ii) As in (i)
No. of lattice points = 4
(iii) In bcc unit cell, number of corner atoms per unit cell
=8 corners*18 per corner atom
=8*18=1 atom
Number of atoms at body centre =1*1 = 1 atom
∴ Total number of atoms (lattice points) = 1 + 1 =
New answer posted
11 months agoContributor-Level 10
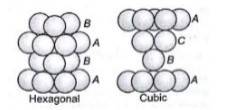
1.31 (i) In hexagonal close packing (hcp), the spheres of the third layer are vertically above the spheres of the first layer. It means tetrahedral voids of the second layer are covered by the spheres of the third layer. The AB. type.In cubic close packing (ccp), the spheres of third layer cover the octahedral voids of second layer. But the spheres of the fourth layers are aligned with those of the first layer. The pattern is ABC. type.
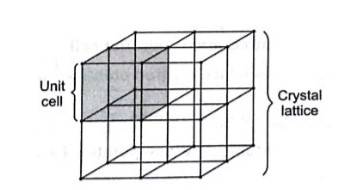
(ii) Crystal lattice is the three dimensional arrangement of identical point in the space which represent how the constituent particles (atoms, ions, molecules) are arranged in a crystal.Unit cell is th
New answer posted
11 months agoContributor-Level 10
(b) The compound contaminates the groundwater and is also a suspected carcinogen.
New answer posted
11 months agoContributor-Level 10
(b) CO is highly poisonous to living beings because of its ability to block the delivery of oxygen to the organs and tissues.
New question posted
11 months agoTaking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers