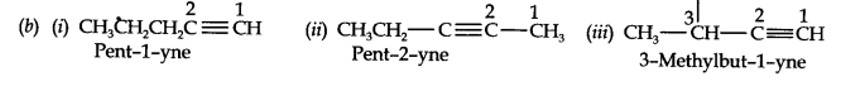Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New question posted
11 months agoNew answer posted
11 months agoContributor-Level 10
1.42 The ratio less than 2:1 in Cu2O shows that some cuprous (Cu+) ions have been replaced by cupric (Cu+2) ions. To maintain electrical neutrality, every two Cu+ ions will be replaced by one Cu+2 ion, thereby creating a hole. As conduction will be due to the presence of these positive holes, hence it is a p -type semi conductor
New answer posted
11 months agoContributor-Level 10
1.41 These solids have conductive in the intermediate range from 10−6 to 104ohm−1m−1. As there is rise in
the temperature, conductivity also increases because electrons from the valence band jump to
conduction band.
Types of semiconductors
(a) n - type semiconductor when silicon or germanium crystal is doped with group 15 element like P or
As, the dopant atom forms four covalent bonds like a Si or Ge atom but the fifth electron no used in
bonding, becomes delocalised and contribute its share towards electrical conduction. Thus, silicon or
germanium doped with P or As is called n-type semiconductor (negative
New answer posted
11 months agoContributor-Level 10
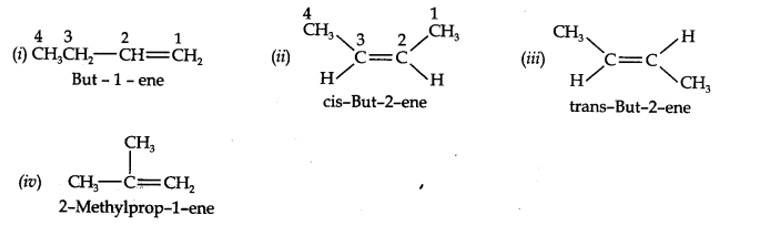
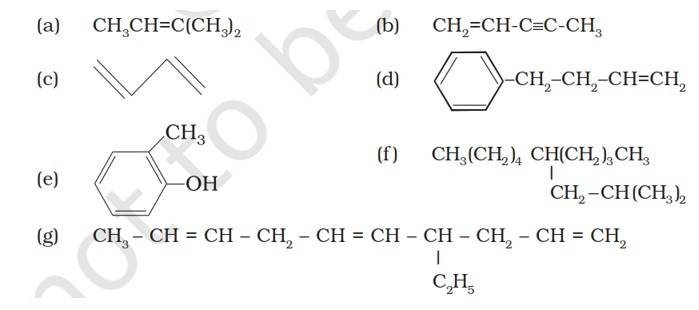
(a) 2-Methykbuut-2-ene
(b) Pent-1-ene-3-yne
(c) But-1,3-diene
(d) 4-Phenylbut-1-ene
(e) 2-Methyl phenol (f) 5- (2-Methylpropyl)decane
(g) 4-Ethyldeca-1,5,8-triene
New answer posted
11 months agoContributor-Level 10
Chlorination of methane is a free radical reaction which occurs by the following mechanism involving initiation, propagation and termination steps:

New answer posted
11 months agoContributor-Level 10
Analysis shows that nickel oxide has the formula Ni0.98 O1.00. What fractions of nickel exist as Ni2+ and Ni3+ ions?
1.40 It is given that nickel oxide has the formula as Ni0.98 O1.00.
As per the formula, there are 98 Ni ions for 100 oxide ions.
Out of 98 Ni ions, let x ions be in +2 oxidation state
98−x ions will be in +3 oxidation state.
Oxide ion has −2 charge.
To maintain electrical neutrality, total positive charge on cations = total negative charge on anions.
2x+3(98−x)+100(−2)=0
x=94
Fraction of Ni2+ ions = 94/98 = 0.96
Fraction of Ni2+ ions = 98-94/98 = 0.04
Hence, the fractions of nickel that exists as Ni2+ and Ni3+ are 0.
New answer posted
11 months agoContributor-Level 10
Analysis shows that nickel oxide has the formula Ni0.98 O1.00. What fractions of nickel exist as Ni2+ and Ni3+ ions?
1.41 It is given that nickel oxide has the formula as Ni0.98 O1.00.
As per the formula, there are 98 Ni ions for 100 oxide ions.
Out of 98 Ni ions, let x ions be in +2 oxidation state
98? x ions will be in +3 oxidation state.
Oxide ion has ?2 charge.
To maintain electrical neutrality, total positive charge on cations = total negative charge on anions.
2x+3 (98? x)+100 (?2)=0
x=94
Fraction of Ni2+ ions = 94/98 = 0.96
Fraction of Ni2+ ions = 98-94/98 = 0.04
Hence, the fractions of nickel that exists as Ni2+ and Ni3+ are 0.96 a
New question posted
11 months agoTaking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers