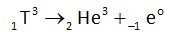Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
6 months agoThe nature of charge on resulting colloidal particles when FeCl3 is added to excess of hot water is:
New answer posted
6 months agoContributor-Level 10
Tritium is radioactive and it decays into He3 during emission of b-radiation
New answer posted
6 months agoContributor-Level 10
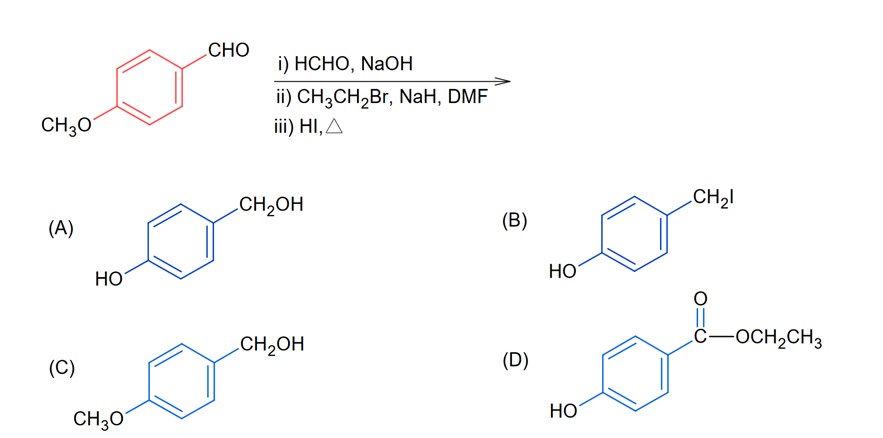
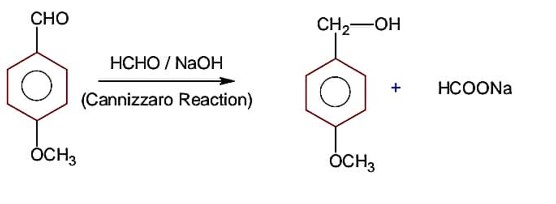
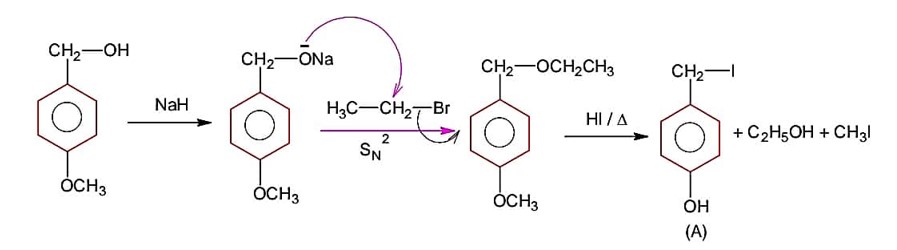
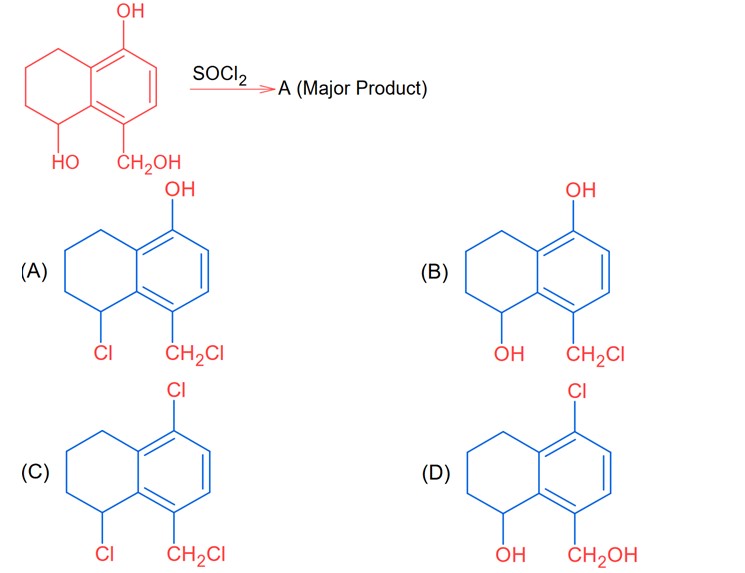
Since SOCl2 is used to covert aliphatic (R-OH) into chlorides. It will not react with aromatic alcohol
New answer posted
6 months agoContributor-Level 10
Siderite – FeCO3 (ore of iron)
Calamine – ZnCO3 (ore of zinc)
Malachite – CuCO3.Cu (OH)2 (ore of copper)
Cryolite – Na3AlF6 (ore of aluminium)
New answer posted
6 months agoContributor-Level 9
It happens in reversible reactions when the rate of the forward reaction becomes equal to the rate of the backward reaction. Result in the same concentration of reactants and product.
New answer posted
6 months agoContributor-Level 9
If the conditions of equilibrium are changed, it shifts to oppose the change. For example, in Haber's process, high pressure favors NH? formation.
New answer posted
6 months agoContributor-Level 9
The equilibrium constant is the ratio of the concentrations of products to reactants, each raised to the power of their stoichiometric coefficients. For reversible reactions.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers