Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
CrO? ²? (Cr? ) → Cr? ³, Total oxidation number change = 3
MnO? (Mn? ) → Mn? ², Total oxidation number change = 5
Cr? O? ²? (Cr? ) → 2Cr? ³, Total oxidation number change = 6
C? O? ²? (C? ³) → 2CO? (C? ), Total oxidation number change = 2
New answer posted
7 months agoContributor-Level 9
B.D.E H-H < B.D.E D-D
B.D.E H-H = 435.9 kJ / mole
B.D.E D-D = 443.4 kJ / mole
E H ≈ E D - 7.5
New answer posted
7 months agoContributor-Level 10
Rotamers or conformers arises due to free rotation along σ - bond.
New answer posted
7 months agoContributor-Level 10
Rutherford atomic model can not explain hydrogen spectrum it is explained by Bohr's atomic model and from Bohr's atomic model, uncertainity principle can't be explained.
New answer posted
7 months agoContributor-Level 10
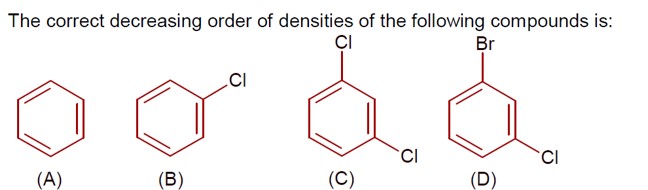
Density ∝ molar mass, as size of given molecules is nearly same
New answer posted
7 months agoContributor-Level 9
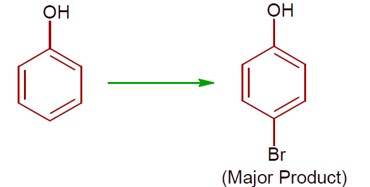
(a) Phenol with Br? / H? O gives 2,4,6-tribromophenol. (High ionisation due to polar solvent and high activation of ring)
(b) Phenol with Br? in CS? /CHCl? /FeBr? gives a mixture of o- and p-bromophenol, with p-bromophenol as the major product. (less ionisation due to non- polar solvent)
[Chemical reactions showing bromination of phenol under different conditions]
New answer posted
7 months agoContributor-Level 10
Bohr's model played an important role in the development of quantum theory. It introduced the idea of quantised electron orbits. It disproved claims of classical mechanics, which predicted that electrons would spiral into the nucleus. Bohr proposed that electrons can exist only in specific, stable energy levels called stationary states. There would be transitions between these levels that would help Bohr explain the line spectra of hydrogen. That together linked the atomic structure with the concept of energy quantisation. Even though the model could not explain multi-electron atoms, it laid the foundation for modern quantum mechanics.
New answer posted
7 months agoContributor-Level 10
The existence of atomic spectra tells us that energy levels in atoms are quantised. When atoms absorb or emit light, they do so at specific wavelengths. They lead to line spectra instead of a continuous spectrum. Now, every line corresponds to an electron that transitions between fixed energy levels. This is to make the photon's energy equal to the difference between them. If energy levels were not discrete, the spectra would be continuous. So, the line spectra provide direct evidence that electrons in atoms occupy quantised energy states.
New answer posted
7 months agoContributor-Level 10
Metal of group 7, 8, & 9 dose not form interstitial hydride this is called hydride gap.
Mn → group - 7
Fe → group - 8
Co → group - 9
So, Cr will forms interstitial hydride.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers