Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
This is a Long Answer Type Question as classified in NCERT Exemplar
Explanation- speed of jackets = 125m/s
Height of hill = 500m
To cross the hill vertical component of velocity should be grater than this value uy=
So u2= ux2+uy2
Horizontal component of initial velocity ux =
Time taken to reach the top of hill t=
Time taken to reach the ground in 10 sec = 75 (10)= 750m
Distance through which the canon has to be moved =800-750=50m
Speed with which canon can move = 2m/s
Time taken canon = 50/2= 25s
Total time t= 25+10+10= 45s
New answer posted
9 months agoContributor-Level 10
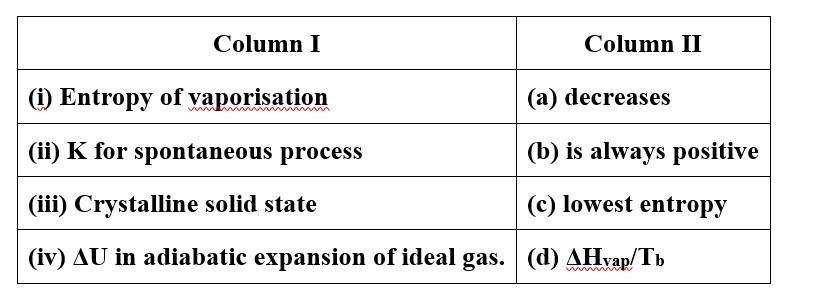
This is a Matching Type Questions as classified in NCERT Exemplar
(i)- (b), (d)
(ii)- (b)
(iii)- (c)
(iv)- (a)
New answer posted
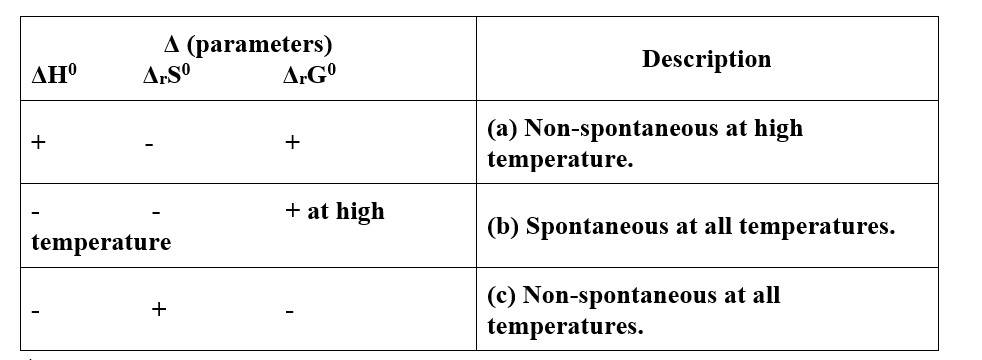
9 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
(i)- (c)
(ii)- (a)
(iii)- (b)
New answer posted
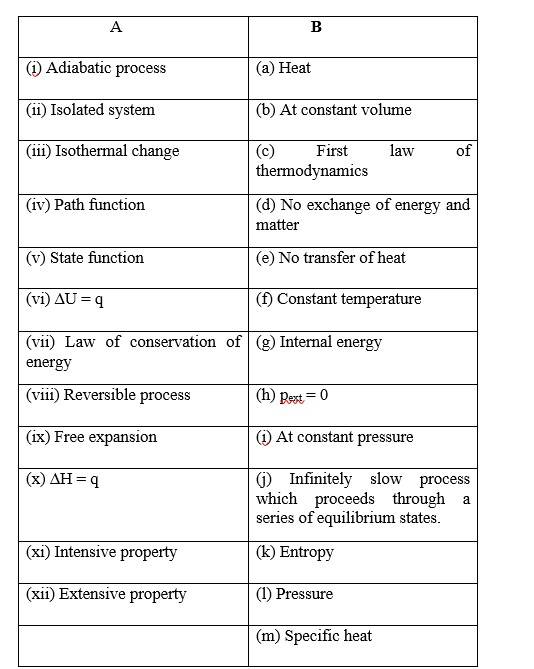
9 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
(i) (b)
(ii) (c)
(iii) (a)
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
option (i) and (iii)
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
option (iv)
For
isothermal reversible change
q= -w = nRTln =2.303nRTlog
= = =2
For
isothermal expansion of ideal gases, ? U = 0
Since,
temperature is constant this means there is no change in internal energy.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
option (iii) and (iv)
Gas expands to fill the available space spontaneously, and burning of carbon to carbon dioxide is spontaneous
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
option (i) and (ii)
In an exothermic reaction, heat is evolved, and system loses heat to the surroundings. Therefore, qp will be negative and? rH will also be negative. Hence, option (i) and (ii) is the correct answer.
New answer posted
9 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
option (i)
The laws of thermodynamics deal with energy changes of macroscopic systems involving a large number of molecules rather than microscopic systems containing a few molecules. Laws of thermodynamics apply only when a system is in equilibrium or moves from one equilibrium state to another equilibrium state.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers