Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
m =50g = 50
Side = 1cm = 0.01m
Speed v = 0.1m/s
Young's modulus= 2 2
According to the formula
F/A= Y
And F= K where K is the compression in the spring.
K= YA/L = YL
Initial KE= 2 (1/2mv2)= 5
Final PE= 2 (1/2)K ( )2
=
New answer posted
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
Let M be the mass of the rocket at any time t and v1 the velocity of the rocket at the same time t
Let? m = mass of gas ejected in? t time
Relative speed of the gas ejected =u
KE +? t = KE of rocket +KE of gas
= ½ (M-? m) (v+? v)2 + ½? m (v-u)2
KEt= KE of rocket at time t= ½ Mv2
So? K = KEt+? t -KEt
= (M? v=? mu)v+1/2? mu2
Since action and reaction forces are equal
M? v/? t=? m/? t|u|
M? v=? m u
So? K= ½? mu2
? K=? W
? W=1/2? mu2
New answer posted
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
(a) As ball 1 is rolling down without slipping there is no dissipation of energy hence, total mechanical energy is conserved. Ball 3 is having negligible friction hence, there is no loss of energy.
(b) Ball 1 acquires rotational energy, ball 2 loses energy by friction. They cannot cross at C. Ball 3 can cross over.
(c) Ball 1, 2 turn back before reaching C. Because of loss of energy, ball 2 cannot reach back to A. Ball 1 has a rotational motion in “wrong” sense when it reaches B. It cannot roll back to A, because of kinetic friction.
New answer posted
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
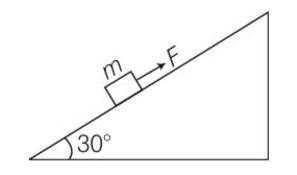
(a) work done= increase in PE
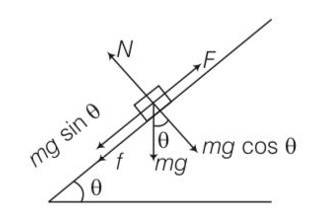
= mg (vertical distance travelled)
= mg (s)sin = 50J
(b) work done against friction = fs
=
= 0.1
(c) increase in PE =mgh
=1
(d) according to work energy theorem W= change in KE
= -mgh-fs+FS
= -50-8.66+10 (10)
= 41.34J
(e) force f = FS
= 10 (10)= 100J
New answer posted
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
i d + 48
ii a &n
New question posted
10 months agoNew answer posted
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
i ) b 1681 3374 – 328
ii ) a 419 3051 – 48
iii)
New answer posted
10 months agoContributor-Level 10
This is a Matching Type Questions as classified in NCERT Exemplar
Be 111
C
New answer posted
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Option (ii)
The electron gain enthalpy decreases down the group due to the increase in the atomic size which leads to decrease in the effective nuclear charge.
New answer posted
10 months agoContributor-Level 10
This is a Assertion and Reason Type Questions as classified in NCERT Exemplar
Option (iii)
The Be has the electronic configuration of [He]2s2 while B has the electronic configuration of [He]2s22p1 . As in Be the outermost orbital i.e. 2s is fully filled thus it provides extra stability to the Be compared to B. Apart from this 2s electron is more penetrated compared to the 2p electron for which 2p electron faces more shielding effect than that of 2s electron.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers