Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Long Answers Type Questions as classified in NCERT Exemplar
In BF3, due to n−pπ back bonding between the vacant p-orbital of boron and filled p-orbital of fluorine. This Π− pπ back bonding is absent in case of hydrogen as it is a single electron element.
Two BH3 molecules dimerise to form diborane.
In B2H6 There are two types of hydrogens present.
(I) Four hydrogens that are terminally bonded to each of two boron atoms.
(II) Two hydrogens that are bonded to both boron atoms forming a bridge in between.
The four terminal hydrogen atoms and two boron atoms lie in the same plane while bridging hydrogen lies in a plane perpend
New answer posted
10 months agoContributor-Level 10
This is a Long Answers Type Questions as classified in NCERT Exemplar
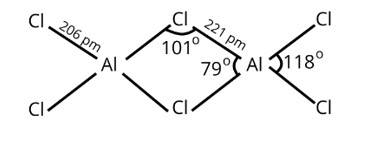
Both the compounds, BCl3 and AlCl3 are electron deficient compounds. In BCl3, boron is smaller in size and cannot assemble four big chlorine atoms near it causing steric hindrance and making it unstable.
Hence, BCl3 exists as a monomer only.
In AlCl3, aluminum has 3p-orbitals through which chlorine atoms can be accommodated easily to complete its octet and dimer is formed.
New answer posted
10 months agoContributor-Level 10
(i) TlCl is more stable than TlCl3, due to inert pair effect, +1 oxile. oxidation state.
(ii) AlCl3, Al3+ is more stable than aluminum ions in +1 state.
(iii) Due to the inert pair effect, +1 oxidation state is more stable than the +3 oxidation state. So, InCl is more stable than InCl3.
(iv) Boranes correspond to alkane-like compounds of boron. They consist of boron and hydrogen. Most common borane existing is dibecane.
4BF3 + 3LiAlH4?2B2H6 + 3LiF + 3AIF3
New answer posted
10 months agoContributor-Level 10
This is a Long Answers Type Questions as classified in NCERT Exemplar
When aqueous solution of borax is acidified with hydrochloric acid, boric acid is produced. As the name says, boric acid is acidic in nature but weak acid. Unlike protonic acid, boric acid is monobasic acid. It accepts electrons from the hydroxyl group of water and forms [B (OH)4]-.
New answer posted
10 months agoContributor-Level 10
This is a Long Answers Type Questions as classified in NCERT Exemplar
When aqueous solution of borax is acidified with hydrochloric acid, boric acid is produced. As the name says, boric acid is acidic in nature but weak acid. Unlike protonic acid, boric acid is monobasic acid. It accepts electrons from the hydroxyl group of water and forms [B (OH)4]-.
New answer posted
10 months agoContributor-Level 10
This is a assertion and reason type question as classified in NCERT Exemplar
(A) Explanation:
Alkaline earth metals are stable at room temperature except beryllium carbonate. It decomposes to give beryllium oxide and carbon dioxide. It is kept in an atmosphere of carbon dioxide so that equilibrium shifts to the right. The reaction is shown below. BeCO3 → BeO+CO2
New answer posted
10 months agoContributor-Level 10
This is a assertion and reason type question as classified in NCERT Exemplar
(A) Lithium beimg small in size, polarises large carbonate ion. Polarisation is the distortion of electron cloud of the anion by the cation. Thus, the carbonate of lithium decomposes easily on heating to form lithium oxide and CO2. The reaction is shown below.
Li2CO3 → Li2O+CO2
New answer posted
10 months agoContributor-Level 10
This is a Long Answers Type Questions as classified in NCERT Exemplar
(i) ln? AlCl3, Aluminium is bonded to 3 chlorine atoms through three covalent bonds and six electrons are shared in the structure. To have complete Octet, AlCl3 lacks two electrons & hence act as an electron acceptor substance or Lewis acid.
(ii) Undoubtedly, fluorine has more electronegativity than chlorine, BF3 is stronger Lewis acid than BCl3 because of n−p π back bonding in BF3, both the constituting atoms boron and fluorine are involve p-orbital in back bonding. On moving down the group, the size of halogen atoms increases, back bonding decreases and Lewis ac
New answer posted
10 months agoContributor-Level 10
This is a matching type question as classified in NCERT Exemplar
(i) → (f); (ii) → (d); (iii) → (b) : (iv) → (c) ; (v) → (e); (vi) → (a)
All alkali metal and alkaline earth metals except beryllium and magnesium gives characteristic color when introduced into flame. Due to released energy being absorbed in the visible region, alkali metals and alkaline earth metals gives characteristic color.
(i) Caesium imparts blue color when introduced into flame.
(ii) Sodium imparts yellow color when introduced into flame.
(iii) Potassium imparts violet color when introduced into flame.
(iv) Calcium imparts brick red color when introduced
New answer posted
10 months agoContributor-Level 10
This is a matching type question as classified in NCERT Exemplar
(i) → (c); (ii) → (d); (iii) → (b) : (iv) → (a)
(i) CaCO3 is used in manufacturing of high quality paper.
(ii) Due to the disinfectant nature of Ca (OH)2 is used in white wash.
(iii) CaO is used in the manufacture of sodium carbonate from caustic soda.
(iv)CaSO4 is used in dentistry, ornamental work and for making statues.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers
