Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
C
Boric acid is Lewis acid, having six electrons in its valence shell. It combines with water, accepts electrons
from OH−of water molecule and complete it octet to 8 and releases H+
Reaction: B (OH)3 + OH − H→ [B (OH)4]− + H+
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Let b be the position vector of the centre of mass of a regular n-polygon.
(n-1) equal point masses are placed at (n-1) vertices of the regular n polygon, therefore for its centre of mass
rCM=
(n-1)mb+ma=0
B=-
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
A
Number of items/molecules bonded to the central atom is termed as coordination number. In the given MF63−
Coordination number of metal is six and boron can have topmost coordination number of four as it consists of s and p orbital only and lacks d-orbital. Therefore, boron cannot be compared in the form of MF63−.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
A
The B2O3 reacts with water to form boric acid hence it is an acidic oxide. As we move down the group electronegativity decreases and the tendency to donate electrons increases therefore basic character increases or acidic character decreases. So, from the given options, the most acidic oxide is B2O3.
New answer posted
10 months agoContributor-Level 10
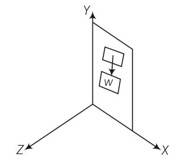
This is a short answer type question as classified in NCERT Exemplar
Where weight of the door acts along negative y axis
A force can produce torque only along a direction normal to itself as . So when the door is in the xy plane the torque produced by gravity can only along z direction. Never about an axis passing through y direction. Hence the weight will not produce any torque.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
A
Central atom in [B (OH)4 ]− no lone pair. Electronic conf In ground state: 1s2 2s2 2px1 2py0 2pz0
In excited state, One Electron from 2s shifts to 2py orbital and the configuration becomes:
1s2 2s2 2p1 x2 p1 y2 pz0
Now, one s and three p orbitals combined to give sp3 hybridisation and tetrahedral shape.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
A
Lewis acids are the species in which the state is not complete and ready to accept electrons. Because Al is surrounded by 6 electrons in AlCl3 and all three Cl atoms are surrounded by 8 electrons, AlCl3 is an electron acceptor. It is a covalent compound.
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Wheel is a rigid body. The particles that constitute the wheel do experience a centripetal acceleration directed towards the centre. This acceleration arises due to internal elastic forces which cancel out in pairs.
In a half wheel the distribution of mass about its centre of mass is not symmetrical, therefore the direction of angular momentum of the wheel does not coincide with the direction of its angular velocity. Hence an external torque is required to maintain the motion of the wheel.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
C
Gallium generally exists as solid at room temperature but melts on slight heating (melting point: 20? C ).
Whereas the boiling point of Gallium is very high around 2400? C. Gallium has large cohesive forces that hold its structure together and it is stable for a wide range of temperatures and can be used for measuring high temperatures.
New answer posted
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
4BF3 + 3LiAlH4? 2 B2H6 + 3LiF + 3AlF3
(Z) (X)
B2H6 + 6H2O?2H3BO3 + 6H2
(X) (Y)
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers