Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
H3BO3
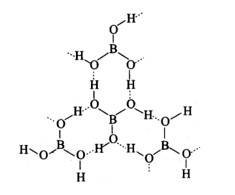
Boric acid forms a hexagon and rings through hydrogen bonding and has a layer-like structure.
Boric acid is present in water as [B (OH)4]−.H3BO3 electron from the OH of water and forms the complex BOH for negative for sp3 and is present in sp3 hybridisation.
Reaction:
B (OH)3+2H2O→ [B (OH)4]− + H3O+
New answer posted
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
(a) Before being bought in contact with the table the disc was in pure rotational motion. Hence Vcm=0
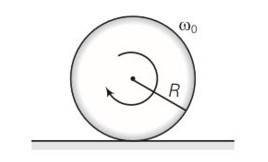
(b) when the disc is placed in contact with table due to friction centre of mass acquires some linear velocity.
(c) when the rotating disc is placed in contact with the table due to friction centre of mass acquires some linear velocity.
(d) friction is responsible for the effects ib b and c
(e) when rolling starts Vcm=wR
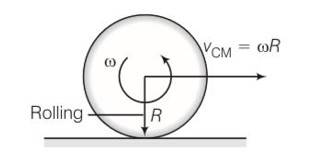
(f) time period for rolling to begin is t=
New answer posted
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
(a) yes the law of conservation of angular momentum can be applied because there is no net external torque on the system of the two discs.
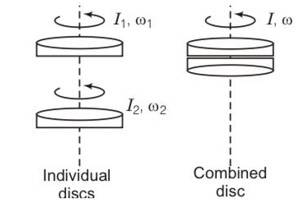
External forces gravitation and normal reaction act through the axis of rotation, hence produce no torque.
(b) by conservation of angular momentum
Lf= Li
Iw=I1w1+I2w2
So w=
(c) Kf=
Kf= ½ (I1w12+I2w22)
Kf-Ki =- 2<0
(d) hence there is loss of KE of the system. The loss of kinetic energy is mainly due to work against the friction.
New answer posted
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
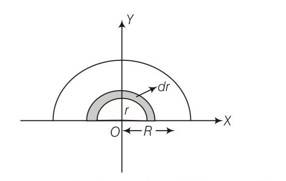
let M and R be mass and radius of half disc , mass per unit area of half disc
So m =
(a) the half disc be supposed to be consist of a large number of semicircular ring of mass dm and thickness dr and radii ranging from r=0 to r=R.
surface area of semicircular ring of radius r and thickness dr =
so mass of the elementary ring dm =
dm=
if x,y are coordinates of centre of mass of this element,
then (x,y)=(0,2r/ )
so x=0 and y =2r/
let xcm and ycm be the coordinates of the centre of mass of the semicircular disc
so xcm=
Ycm=
= 0R
= 4R/3
Centre of mass of a uniform quar
New answer posted
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
H3BO3 or B (OH)3 is an electron deficient compound or Lewis acid which easily accepts electron from OH of water and releases its proton hence it is a Monobasic acid.
Reaction:
B (OH)3 + 2H2O → [B (OH)4]− + H3O+
New answer posted
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
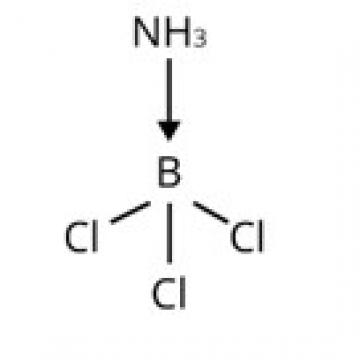
The central B atom in BCl3 has six electrons in its valence shell. As a result, it is an electron-deficient molecule in need of two more electrons to complete its octet. To put it another way, BCl3 acts as a Lewis acid. NH3 on the other hand, has a lone pair of electrons that it can easily donate. As a result, NH3 serves as a Lewis base. As shown below, the Lewis acid (BCl3) and Lewis base (NH3) combine to form an adduct:
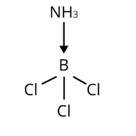
The valence shell of AlCl3 contains six electrons. As a result, it is an electron-deficient molecule that requires two additional electrons to compl
New answer posted
10 months agoContributor-Level 10
This is a Long Answers Type Questions as classified in NCERT Exemplar
2C (s) + O2 + 4N2 (g) ![]() 2CO (g) + 4N2 ( g)
2CO (g) + 4N2 ( g)
Fe2O3 (S)+3CO (g) ![]() 2Fe (S)+3CO2
2Fe (S)+3CO2
C= tetravalent carbon
CO= carbon monoxide
Fe2O3= ferric oxide
CO2= carbon dioxide
Tetravalent elements i.e. carbon combines with oxygen to produce carbon monoxide. The reaction occurs at high temperatures and in the presence of nitrogen gas, which acts as a producer gas only. It is not consumed in the reaction. The carbon monoxide formed acts as a reducing agent for ferric oxide, reduces the oxidation state of iron from +3 to zero, and oxidiz
New answer posted
10 months agoContributor-Level 10
This is a Long Answers Type Questions as classified in NCERT Exemplar
Boron is the only non-metallic and extremely hard element in group 13, and it is also used to make bulletproof vests. Boron exists in a variety of allotropic forms. It usually has a high melting point and no d orbital. Using 2s and 2p orbitals, it can achieve a maximum covalency of 4 . Because the octet of boron is not completed in trivalent halides of boron, it acts as Lewis acid. It forms an adduct when it reacts with Lewis base.
BF3+NH3→H3N−B−F3
New answer posted
10 months agoContributor-Level 10
This is a Long Answers Type Questions as classified in NCERT Exemplar
B2H6 + 2NMe3→ 2BH3NMe3 BH3NMe3+H2O → H3BO3 + NMe3 + 6H2
(A) (B) (C)
(A): B2H6
(B): 2BH3NMe3
(C): H3BO3
New answer posted
10 months agoContributor-Level 10
This is a Long Answers Type Questions as classified in NCERT Exemplar
(i) Organosilicon polymers having (R2SiO2) as monomer units are called silicones. Silicones contain organic side groups which surround it giving alkane like nature and makes it hydrophobic. Silicones are applicative in electrical insulators, water proofing, sealant. They have been utilized in the biological field in cosmetic implants and other surgeries.
(ii) Boranes correspond to alkane-like compounds of boron. They consist of boron and hydrogen. Most common borane existing is dibecane.
4BF3 + 3LiAlH4?2B2H6 + 3LiF + 3AIF3
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers