Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
When a trivalent atom is added to the crystal of SiO2, it substitutes silicon atoms which result in generation of holes. These holes make the crystal conductor of electricity. The overall Crystal is electrically neutral and is called the p-type conductor.
New answer posted
10 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
When the vertical height of the objects is very small as compared to the earth's radius we call the objects small, otherwise it is extended. Building and ponds are small objects and deep lake and ocean are examples of extended objects.
New answer posted
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
As compared to carbon, silicon is bigger in size and is less electronegative. It shows resistance in forming p−p multiple bonding which is easily done by carbon. Thus, SiO2 is a 3−D network where each silicon is linked covalently to 4 oxygen atoms while in CO2, Carbon is linked with two oxygen atoms with double bond in a linear manner.
New answer posted
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
As we move down the group in group 13 and 14 the participation of s-electrons in bond formation decreases the primary reason behind this is the inert pair effect.
In this the p-electrons take part in bond formation and more energy is required to unpack the valence electrons to make them participate in bonding. Due to this the lower oxidation state of elements becomes stable done the hire oxidation state. As for group 13, +1 oxidation state is more stable than +3 and for group 14, +2 oxidation state is more stable than +4.
New answer posted
10 months agoContributor-Level 10
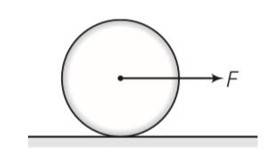
This is a long answer type question as classified in NCERT Exemplar
Frictional force f is acting in the opposite direction of F . let the acceleration of centre of mass of disc be a then
F-f=Ma where M is the mass of the disc
fR= (1/2 MR2)
so fR= (1/2MR2) (a/R)
Ma=2f
From the above equation
F = F/3
F< =
f/3 <
F=3
New answer posted
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
(A) Carbon has small size and large electronegativity, it forms strong n−pπ bonding with two oxygen atoms forming a separate CO2 molecule.
While in SiO2 silicon is larger in size with comparatively less electronegativity than carbon it shows no tendency to form n−pπ bonding rather forms Single covalent bond with oxygen. Thus, SiO2 possess 3D network-like structure in which each Silicon is bonded to 4 oxygen atoms.
(B) Carbon is smaller in size and lacks d-orbitals hence can have a maximum coordination number of four and sp3 hybridisation only.
Wherea
New answer posted
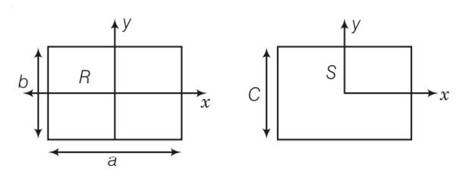
10 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
area of square = area of rectangular plate
C2=a b
(a)
here bxR
(b) here a>c so IyR>IyS
(c) IzR-IzS +2ab= (a-b)2
(IzR-IzS)>0
New answer posted
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
(A) sarbon in CCl4 does not have a vacant d-orbital to accommodate the electrons from OH of water molecules. Also CCl4 is nonpolar covalent compounds whereas H2O is polar. So, no strong interaction occurs between them. Hence CCl4 is miscible in water.
Whereas in SiCl4, silicon has bigger size than carbon and have d-orbitals for accommodation of electrons donated by OH of water in the process of hydroxylation. This leads to a strong interaction and silicon acid Is formed as a product. SiCl4 is completely miscible in water.
(B) As we move from carbon to sil
New answer posted
10 months agoContributor-Level 10
This is a Short Answers Type Questions as classified in NCERT Exemplar
Both BCl3 and AlCl3 are electron deficient compounds that are central atom boron and aluminium have incomplete Octet. In each compound, a metal atom is surrounded by six electrons of three covalent bonds with 3 chlorine atoms.
Each chlorine atom has a complete Octet of eight electrons. The electron deficient compounds act as Lewis acid and readily accept two electrons to complete their octet.
New answer posted
10 months agoContributor-Level 10
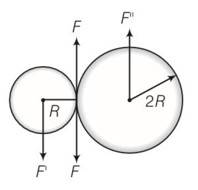
This is a long answer type question as classified in NCERT Exemplar
(a) Situation as given below
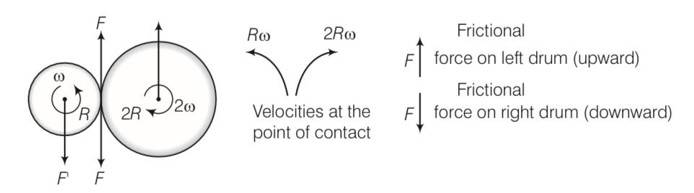
(b) F' =F=F'' where F' and F'' are external forces through support.
So Fnet=O
External torque = F (anticlockwise)
(c) Let w1 and w2 be the final angular velocities of smaller and bigger drum in both clockwise and anticlockwise . finally there will be no friction
hence Rw1=2Rw2
so
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers