Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
velocity of a freely falling body is v=
And
-1
The wavelength of a photon needed to remove a proton from a nucleus which is bound to the nucleus with 1 MeV energy is nearly
a) 1.2 nm (b) 1.2 x 10-3 nm
(c) 1.2 x 10-6 nm (d). 1.2 x 10 nm
New question posted
11 months agoNew answer posted
11 months agoContributor-Level 10
(d)
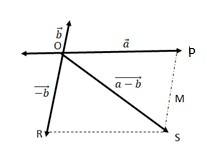
We can write
OS + PS > OP …….(i)
OS > PS – OP …….(ii)
+ ….(iii)
The quantity on the LHS is always positive and that on the RHS can be positive or negative. To make both quantities positive, we take modulus of both sides as:
> ….(iv)
If the two vectors and act along a straight line but in the opposite direction, then we can write = -
Combining (iv) and (v), we get
-
New question posted
11 months agoNew answer posted
11 months agoContributor-Level 10
4.12. No, these cannot be taken as canonical forms because the positions of atoms have been changed.
New answer posted
11 months agoContributor-Level 10
4.11.

Resonance in CO32-, I, II and III represent the three canonical forms.
In these structures, the position of nuclei is the same.
The Lewis dot structure has two single bonds and one double bond.
All three forms have almost equal energy.
The three forms have same number of paired and unpaired electrons; they differ only in their position.
New answer posted
11 months agoContributor-Level 10
4.10. Bond-length: It is the equilibrium distance between the nuclei of two bonded atoms in a molecule. Bond-lengths are measured by spectroscopic methods.
New answer posted
11 months agoContributor-Level 10
4.9. Bond strength is directly proportional to the bond order. Greater the bond order more is the bond strength.
New answer posted
11 months agoContributor-Level 10
4.8. Because of two lone pairs of electrons on O-atom, repulsion on bond pairs is greater in H2O in comparison to NH3. Thus, the bond angle is less in H2O molecules.
New answer posted
11 months agoContributor-Level 10
4.7
Molecule | Number of electron pairs around central atom | Type |
| Molecular geometry | Bond angles |
BeCl2? | 2 | AB2 |
| Linear | 180o |
BCl3? | 3 | AB3 |
| trigonal planar | 120o |
SiCl4? | 4 | AB4 |
| tetrahedral | 109.5o |
AsF5? | 5 | AB5 |
| trigonal bipyramidal | three 120 o, two 90 o |
H2?S | 6 | AB2L2 |
| V-shaped/bent | 92 o |
PH3? | 5 | AB3L |
| trigonal pyramidal | 93.5o |
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers

