Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
4.32.
Sigma bond | Pi bond |
It is formed by axial overlap of the atomic orbitals. | It is formed by the sidewise overlap of the atomic orbitals. |
The bond is quite strong. | It is a comparatively weaker bond. |
Only one lobe of the p-orbitals is involved in the overlap. | Both lobes of the p-orbitals are involved in the overlap. |
Electron cloud of the molecular orbital is symmetrical around the inter-nuclear axis. | The electron cloud is not symmetrical |
New answer posted
11 months agoContributor-Level 10
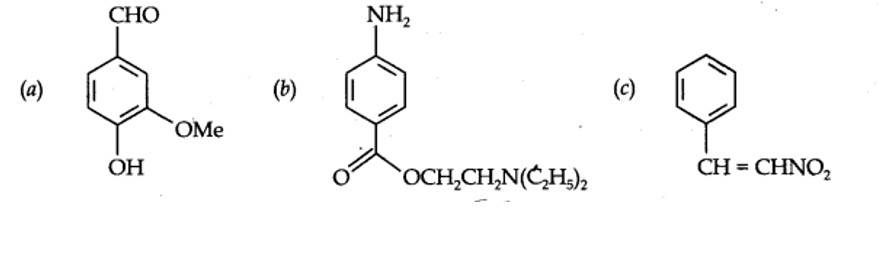
(a) Nucleophilic substitution (b) Electrophilic addition
(c) Bimolecular elimination (d) Nucleophilic substitution with rearrangement.
New answer posted
11 months agoContributor-Level 10
4.31. The electron pair involved in sharing between two atoms during covalent bonding is called shared pair or bond pair. At the same time, the electron pair which is not involved in sharing is called lone pair of electrons.
For example: CH4 has 4 bond pairs but H2O has 2 bond pairs and 2 lone pairs.

New answer posted
11 months agoContributor-Level 10
(a) Here, HO– acts as a nucleophile as it is an electron-rich species, i.e., it is a nucleus-seeking species.
(b) Here, –CN acts as a nucleophile as it is an electron-rich species, i.e., it is a nucleus-seeking species.
(c) CH3C+O acts as an electrophile as it is an electron deficient species.
New answer posted
11 months agoContributor-Level 10
Electrophiles: The name electrophiles mean electron loving. Electrophiles are electron deficient. They may be positive ions or neutral molecules.
Ex: H+, Cl+, Br+, NO2+, R3C+, RN2+, AlCl3, BF3
Nucleophiles: The name nucleophiles means 'nucleus loving' and indicates that it attacks the region of low electron density (positive centres) in a substrate molecule. They are electron rich they may be negative ions or neutral molecules.
Ex: Cl– Br–, CN–, OH–, RCR2–, NH3, RNH2, H2O, ROH etc.
New answer posted
11 months agoContributor-Level 10
Due to hyperconjugation, alkyl groups act as electron donors when attached to a π - system as shown below:

New question posted
11 months agoNew answer posted
11 months agoContributor-Level 10
Nitroethoxide ion (O2NCH2CH2O–) is more stable than ethoxide ion (CH3CH2O–) due to -I effect of nitro group which decreases the negative charge of oxide ion of nitroethoxide ion leading to stability. In contrast, CH3CH2 has +I-effect. It, therefore, tends to intensify the -ve charge and hence destabilizes it.
New answer posted
11 months agoContributor-Level 10
4.29 (c) It will not form a sigma bond because taking x-axis as the inter-nuclear axis, there will be lateral overlap between the two 2py orbitals forming a -bond.
New question posted
11 months agoTaking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers