Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
Depletion of ozone layer creates some sort of holes in the blanket of ozone which surrounds us, this is known as ozone hole.
The consequences are:
- With the depletion of ozone layer, more UV radiation filters into troposphere. UV radiations lead to ageing of skin, cataract, sunburn, skin cancer, killing of many phytoplanktons, damage to fish productivity etc.
- It has also been reported that plant proteins get easily affected by UV radiations which leads to the harmful mutation of cells.
- It also increases evaporation of surface water through the stomata of the leaves and decreases the moisture content of the soil.
4. Increa
New answer posted
11 months agoContributor-Level 10
3.30. (i) For n = 3, the element belong to 3rd period, p-block element.
The electronic configuration is =1s2 2s2 2p6 3s2 3p4. The element name is sulphur.
(ii) For n = 4, the element belongs to 4th period and since the valence shell has 4 electrons it belongs to group 4.
Electronic configuration= 1s2 2s2 2p6 3s2 3p6 3d2 4s2, and the element name is Titanium (Ti).
(iii) For n = 6, the element belongs to 6th period. Last electron goes to the f-orbital, element is from f-block. It belongs to group = 3
The element is gadolinium (z = 64)
Complete electronic configuration = [Xe] 4 f7 5
New answer posted
11 months agoContributor-Level 10
3.29. (i) s-Block elements: ns1-2 where n = 2-7.
(ii) p-Block elements: ns2 np1-6 where n = 2-6.
(iii) d-Block elements: (n-1)d1-10 ns 0-2 where n = 4-7.
(iv) f-Block elements: (n-2)f0-14 (n-1)d0-1 ns2where n = 6-7.
New answer posted
11 months agoContributor-Level 10
3.28. The elements of Group I have only one electron in their respective valence shells and thus have a strong tendency to lose this electron. The tendency to lose electrons in turn, depends upon the ionization enthalpy. Since the ionization enthalpy decreases down the group therefore, the reactivity of group 1 elements increases in the order Li < Na < K < Rb < Cs. In contrast, the elements of group 17 have seven electrons in their respective valence shells and thus have strong tendency to accept one more electron to make stable configuration. So for group 17, the electron gain enthalpy and electronegativity decreases down the group and thus the reactivity also decreases.
New answer posted
11 months agoContributor-Level 10
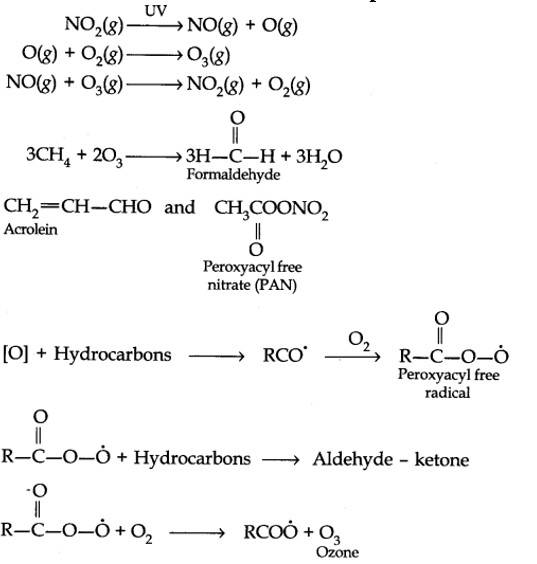
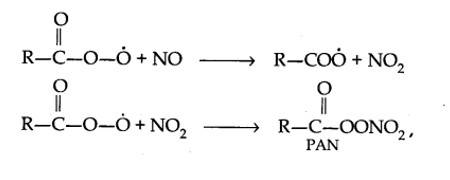
Harmful effects of photochemical smog: Photochemical smog causes serious health problems. Both ozone and PAN (peroxyacetyl nitrate) act as powerful eye irritants. Ozone and nitric oxide irritate the nose and throat and their high concentration causes headache, chest pain, dryness of the throat, cough and difficulty in breathing. Photochemical smog leads to cracking of rubber and extensive damage to plant life. It also causes corrosion of metals, stones, building materials, rubber and painted surfaces.
Control:
- Control of primary precursors of photochemical smog, such as NO2 and hydrocarbons, the secondary precursors such as ozone and PAN
New answer posted
11 months agoContributor-Level 10
3.27. (a) Element belonging to nitrogen family (group 15) e.g., nitrogen.
(b) Element belonging to alkaline earth family (group 2) e.g., magnesium.
(c) Element belonging to oxygen family (group 16) e.g., oxygen.
New answer posted
11 months agoContributor-Level 10
The word smog is a combination of smoke and fog. It is a type of air pollution that occurs in many cities throughout the world. Classical smog occurs in a cool, humid climate. It is also called reducing smog. Whereas photochemical smog (photo means light) occurs in warm and dry sunny climates. It has a high concentration of oxidising agents and therefore, it is also called oxidising smog.
New answer posted
11 months agoContributor-Level 10
This is mainly due to the large number of industries and power plants in the nearby areas. Acid rain has vapours of sulphuric acid dissolved in it. When it comes in contact with various statues or monuments, the acid reacts chemically with marble (calcium carbonate).
CaCO3 + H2SO4 à CaSO4 + H2O + CO2
As a result, the monument is being slowly disfigured, and the marble is getting discoloured and lustreless.
New answer posted
11 months agoContributor-Level 10
3.26.
Metals | Non-metals |
1. They have strong tendency to lose electrons to form cations. | 1. They have strong tendency to accept electrons to form anions. |
2. Metals are strong reducing agents. | 2. They are strong oxidising agents. |
3. Metals have low ionization enthalpies. | 3. Non-metals have high ionization enthalpies. |
4. They form basic oxides and ionic compounds. | 4. They form acidic oxides and covalent compounds. |
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers