Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
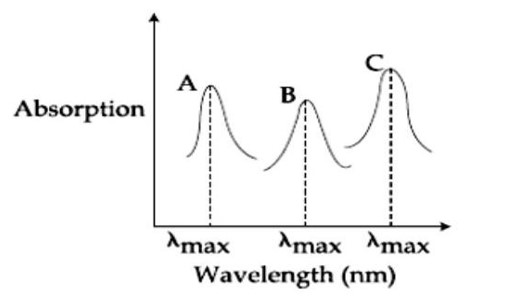
Ligand field strength: NH? > NCS? > F? Stronger ligand, higher Δ, lower λ_max.
So λ (NH? ) < (NCS? ) < (F? ). A= (F? ), B= (NCS? ), C= (NH? ). A-ii, B-i, C-iii.
New answer posted
7 months agoContributor-Level 10
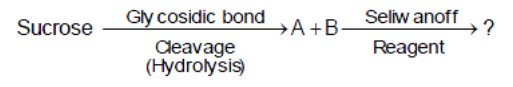
Seliwanoff's test distinguishes aldoses from ketoses. Sucrose hydrolyzes to glucose (aldose) and fructose (ketose). Fructose gives a red color.
New answer posted
7 months agoContributor-Level 10
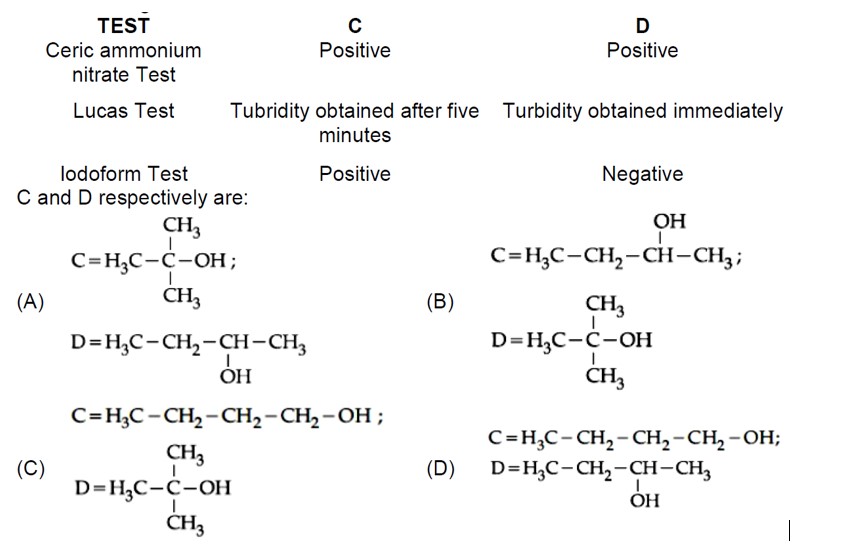
gives iodoform test and slow Lucas test, so it's a methyl secondary alcohol. D gives fast Lucas test, so it's a tertiary alcohol. The Grignard products must be tertiary and secondary alcohols. So A and B must be an aldehyde and a ketone.
New answer posted
7 months agoContributor-Level 10
. [XeF? ]? has 7 electron pairs (5 bonding, 2 lone), pentagonal planar. XeO? F? has 5 electron pairs (trigonal bipyramidal).
New answer posted
7 months agoContributor-Level 9
Sol. (x/m) = k (P)¹/?
log (x/m) = logk + 1/n logP
Slope = 1/n = 2 So n = 1/2
Intercept ⇒ logk = 0.477 So k = Antilog (0.477) = 3
So (x/m) = k (P)¹/? = 3² = 48
New answer posted
7 months agoContributor-Level 9
The oxidation states of iron in these compounds will be
A = +2
B = +4
C = 0
The sum of oxidation states will be = 6.
New answer posted
7 months agoContributor-Level 9
Sol. E? cell = E? (Sn²? |Sn) - E? (Cu²? |Cu)
= -0.16 - 0.34 = -0.50V
ΔG? = -nFE? cell
= -2 * 96500 * (-0.5) = 96500 J
= 96.5 kJ = 96500 J
New answer posted
7 months agoContributor-Level 10
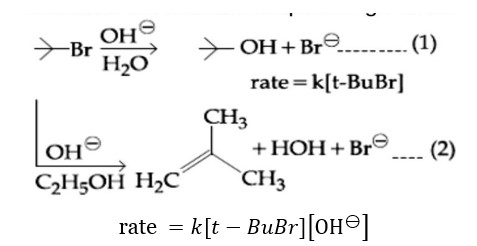
Reaction (1) is SN1 (rate independent of [OH? ]). Reaction (2) is E2 (rate depends on [OH? ]).
Statement (B) is correct. Changing concentration of base will have no effect on reaction (1).
New answer posted
7 months agoContributor-Level 9
Cesium has lowest ionisation enthalpy and hence it can show photoelectric effect to the maximum extent hence it is used in photo electric cell.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers