Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (i)
The orbitals nearer to the nucleus possess more shielding effect.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (iv)
Actinoids possess Z=90-103
Terbium is lanthanoid.
New answer posted
10 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Option (ii)
All the given ions are isoelectronic species thus their radii depend upon the charge more the negative charge higher would be the atomic radii and higher the positive charge lesser would be the atomic radii.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
The electronegativity of the alkali metals decreases down the group due to the increase of the shells thus Cs (Caesium) is the least electronegative metal in the alkali metals.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
The radius of Na+ ion is smaller than that of Na is due to the following reasons:-
(i) The effective nuclear charge of Na+
(ii) The disappearance of 3s orbital from its outermost shell electronic configuration.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Metallic character- Tendency to lose electrons.
Non-Metallic character- Tendency to accept electrons.
Across the period the metallic character decreases whereas non-metallic character increases across the period.
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
(a) As the effective nuclear charge increases and shielding effect decreases across the periods thus the electronegativity increases on moving from left to right in the periodic table.
(b) As the number of shells increases down the group thus its ionisation enthalpy decreases down the group.
New answer posted
10 months agoContributor-Level 10
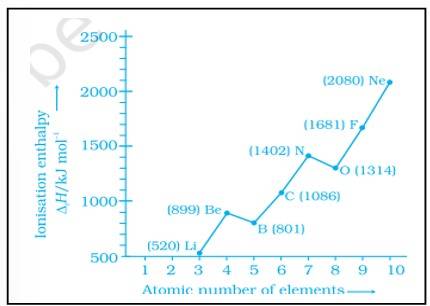
The deviation in the ionization enthalpy of some elements from the general trend can be explained by the points as given below:-
(i) The fully filled and half filled orbital provide extra stability due to the symmetry
(ii) The effective nuclear charge
(iii) The e- - e- repulsion which lead to instability
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
(i) S
The ionization enthalpy increases across the period and decreases down the group. N possesses a half filled 2p orbital which provides it extra stability due to symmetry.
(ii)P
The non-metallic character increases across the period
New answer posted
10 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Exothermic reaction- The reaction in which heat is released
For example- CaO + CO2 → CaCO3 + Heat
Endothermic reaction- The reaction in which heat is absorbed.
For example- 2NH3 + heat → N2 +3H2
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers