Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
1.22. Speed = Distance / Time
Or, Distance = Speed x Time = 3.0 * 108ms–1 x 2.00 ns = 3.0 * 108ms–1 x 2.00 x 10-9 s = 6 x 10-1 m = 0.600 m
New answer posted
11 months agoContributor-Level 10
1.21. (a) Fixing the mass of dinitrogen as 28 g, masses of dioxygen combined will be 32,64, 32 and 80 g in the given four oxides. Theseare in the ratio 1: 2: 1: 5 which is a simple whole number ratio. Hence, the given data obeys the law of multiple proportions.
(b)
(i) 1 km = 103 m and 1 m = 103 mm. So, 1 km = 103 x 103 mm = 106 mm
Now, 1 pm = 10-12 m. So, 1 km = 103 m x 1012 m = 1015 pm
Therefore, 1 km = 106 mm = 1015 pm
(ii) 1 mg = 10-3 g and 1 g = 10-3kg. So, 1 mg = 10-3 x 10-3 kg = 10-6 kg
Now, 1 mg = 10-3 g and 1 g =
Therefore, 1 mg = 10-6 kg = 106 ng
1L = 1000 mL.So 1 mL = 10-3 L.
Now, 1 mL = 1cm3 and 1dm = 10cm. So, 1 mL = 1
New answer posted
11 months agoContributor-Level 10
1.20. The values after round up with three significant figures are:
(i) 34.2
(ii) 10.4
(iii) 0.0460
(iv) 2810
New answer posted
11 months agoContributor-Level 10
1.19. The significant figures in the given values are:
(i) 2 in 0.0025
(ii) 3 in 208
(iii) 4 in 5005
(iv) 3 in 126,000
(v) 4 in 500.0
(vi) 5 in 2.0034
New answer posted
11 months agoContributor-Level 10
1.18. The scientific notation of the given values are:
(i) 4.8*10?3
(ii) 2.34*105
(iii) 8.008*103
(iv) 5.000*102
(v) 6.0012*100
New answer posted
11 months agoContributor-Level 10
1.17. 15 ppm means 15 parts per million i.e.15 in 106
So, % by mass = 15/106 x 100 = 15 x 10-4 = 1.5 x 10-3%
Molality = No. of moles of solute/Mass of solvent in kg
Percent by mass = 1.5 x 10-3 % means 100 g of the sample contain 1.5 x 10-3 g chloroform.
So, 1000 g or 1 kg of the sample will contain 1.5 x 10-3 x 1000/100 = 1.5 x 10-2 g chloroform.
Molar mass of chloroform = 12 + 1 + (3 x 35.5) = 119.5 g/mol
Therefore, molality = 1.5 x 10-2 /119.5 = 1.26 x 10-4m.
New answer posted
11 months agoContributor-Level 10
1.16. Significant figures are meaningful digits which are known with certainty plus one which is estimated or uncertain. The uncertainty in the experimental or the calculated values is indicated by mentioning the number of significant figures
New answer posted
11 months agoContributor-Level 10
1.15. micro = 10–6, deca = 10, mega = 106, giga = 109, femto = 10–15
New answer posted
11 months agoContributor-Level 10
Hydrolysis of aikyltrichlorosilanes gives cross-linked silicones.
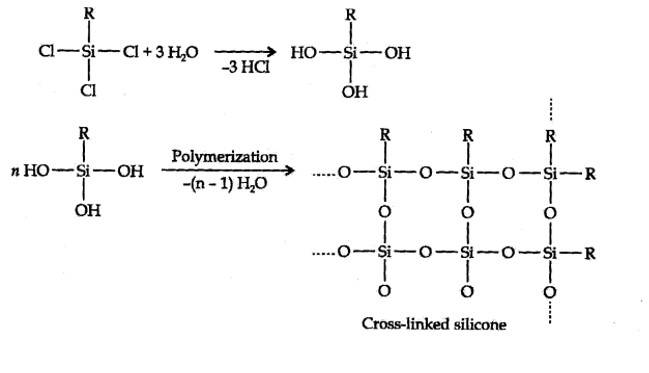
New answer posted
11 months agoContributor-Level 10
Due to inert pair effect, elements of group 14 exhibit oxidation states of +2 and +4. Thus, option (b) is correct.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers
