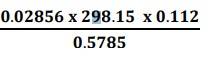Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
1.32. Molar mass of argon is
= [ (35.96755 * 0.337/100)+ (37.96272 * 0.063/100)+ (39.9624 * 99.60/100)]g mol-l
= [0.121+0.024+39.802] g mol-l
= 39.947 g mol-l
So, the molar mass of argon is 39.947 g/ mol.
New answer posted
11 months agoContributor-Level 10
1.31. First we need to find the least precise number to find the significant figures.
(i) Least precise number of the calculation![]() is 0.112
is 0.112
Therefore, number of significant figures in the answer = Number of significant figures in the least precise number, i.e. 3
(ii) Least precise number of calculations = 5.364
Therefore, number of significant figures in the answer will be = Number of significant figures in 5.364 = 4
(iii) 0.0125+0.7864+0.0215
Since the least number of decimal places in each term is four, the number of significant figures in the answer will also be 4.
New answer posted
11 months agoContributor-Level 10
1.30. 1 mol of 12C atoms = 6.02 x 1023 atoms = 12 g
Or, 6.02 x 1023 atoms of 12C have mass = 12 g
Therefore, 1 atom of 12C will have mass = 12 x 1/6.02 x 1023 = 1.9927 x 10-23 g
New answer posted
11 months agoContributor-Level 10
1.29. According to the formula of mole fraction,
X (C2H5OH) = 0.040 = n (ethanol) / [n (ethanol) + n (water)]
Now, n (water) = 1000g/18g mol-1 = 55.55 moles [? Density of water=1kg m-3]
Therefore, 0.040 = n (ethanol) / [n (ethanol) + 55.55]
i.e., 0.04 x n (ethanol) + 2.222 = n (ethanol)
i.e., 2.222 = [1- 0.04] n (ethanol)
i.e., n (ethanol) = 2.222 / 0.96 = 2.314 M
New answer posted
11 months agoContributor-Level 10
1.28. The number of atoms in each of the given elements are calculated as below:
(i) 1 g Au = 1 / 197 mol = 1/ 197 x 6.02 x 1023 atoms
(ii) 1 g Na = 1/ 23 mol = 1/ 23 x 6.02 x 1023 atoms
(iii) 1 g Li = 1/ 7 mol = 1 / 7 x 6.02 x 1023 atoms
(iv) 1 g of Cl2 = 1/ 71 mol = 1/ 71 x 6.02 x 1023 atoms
Therefore, since the denominator is the smallest in case of Li, 1 g Li has the largest number of atoms
New answer posted
11 months agoContributor-Level 10
1.27. (i) 28.7 pm = 28.7 x 10-12 m = 2.87 x 10-11 m
(ii) 15.15 µs = 15.15 x 10-6 s = 1.515 x 10-5 s
(iii) 25365 mg = 25365 mg x 10-6 kg = 2.5365 x 10-2 kg
New answer posted
11 months agoContributor-Level 10
1.26. H2 and O2 react according to the equation
2H2 (g) + O2 (g) ——>2H2O (g)
Thus, 2 volumes of H2 react with 1 volume of O2 to produce 2 volumes of water vapour. Hence, 10 volumes of H2 will react completely with 5 volumes of O2 to produce 10 volumes of water vapour.
New answer posted
11 months agoContributor-Level 10
1.25. Molar mass of Na2CO3= (2 x 23) + 12 + (3 x 16) = 106g mol-1
0.50 mol Na2CO3 means 0.50 x 106 g = 53 g of Na2CO3
0.50 M Na2CO3 means 0.50 mol per volume in litre,
i.e., half of 106 g Na2CO3 is present in 1 L solution.
i.e.,53 g Na2CO3 is present in 1 L of the solution
New answer posted
11 months agoContributor-Level 10
1.24. According to the given equation, 1 mol of N2 reacts with 3 mol of H2.
Or, 28 g of N2 react with 6 g of H2.
So, 2000 g of N2 will react with H2 = 6/28 x 2000 g = 428.6 g of H2.
(i) 2 mol of N2 or 28 g of N2 produce NH3 = 2 mol = 34 g
So, 2000 g of N2 will produce NH3 = 34/28 x 2000 g = 2428.57 g
(ii) Yes, N2 is the limiting reagent while H2 is the excess reagent. So, H2 will remain unreacted.
(iii) H2 will remain unreacted. Mass left unreacted = 1000 g – 428.6 g = 571.4 g
New answer posted
11 months agoContributor-Level 10
1.23. (i) According to the given reaction, 1 atom of A reacts with 1 molecule of B
Thus, 200 molecules of B will react with 200 atoms of A and 100 atoms of A will be left unreacted. Hence, B is the limiting reagent while A is the excess reagent.
(ii) According to the given reaction, 1 mol of A reacts with 1 mol of B
Thus, 2 mol of A will react with 2 mol of B. Hence, A is the limiting reactant since 1 mol of B is left unreacted.
(iii) No limiting reagent.
(iv) 2.5 mol of B will react with 2.5 mol of A. Hence, B is the limiting reagent.
(v) 2.5 mol of A will react with 2.5 mol of B. Hence, A is the limiting reagent
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers