Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
1.9. Average atomic mass = (Fractional abundance of 35Cl x molar mass of 35Cl) + (Fractional abundance of 37Cl x molar mass of 37Cl)
= (75.77/100 x 34.9689) + (24.23/100 x 36.9659)
= 26.4959 + 8.9568
= 35.4527
New answer posted
11 months agoContributor-Level 10
(a) When borax is heated strongly, it loses water and swells into the white mass, which on further heating melts to form a transparent glassy solid called borax glass and borax bead.
Na2B4O710H2O→ Na2B4O7+ 10H2O
Na2B4O7 → 2NaBO2+ B2O3
(b) When boric acid is added to water, it accepts electrons from –OH ion. Boric acid is sparingly soluble in cold water however fairly soluble in hot water.
B (OH)3→ + 2H2→ O→ [B (OH)4→ ]→ + H3→ O+
(c) Al reacts with dilute NaOH to form sodium tetrahydroxoaluminate (III). Hydrogen gas is liberated in the process.
2Al + 2NaOH + 6H2O→ 2Na+ [Al (O
New answer posted
11 months agoContributor-Level 10
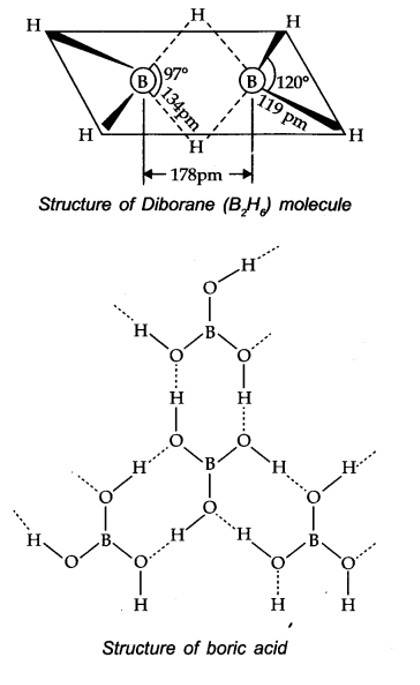
Boric acid contains planar BO33- ions which are linked together through hydrogen bonding shown in the fig.
New answer posted
11 months agoContributor-Level 10
Excess of CO2 absorbs heat radiated by the earth. Some of it is dissipated into the atmosphere while the remaining part is radiated back to the earth. As a result, temperature of the earth increases. This is the cause of global warming.
New answer posted
11 months agoContributor-Level 10
1.7. 1 mole of CuSO4 contains 1 mole (1 g atom) of Cu
Molar mass of CuSO4= 63.5 + 32 + (4 x 16) = 159.5 g mol-1
Thus, Cu that can be obtained from 159.5 g of CuSO4 = 63.5 g
∴ Cu that can be obtained from 100 g of CuSO4 =63.5/159.5 * 100 = 39.81g
New answer posted
11 months agoContributor-Level 10
The highly poisonous nature of CO arises because of its ability to form a complex with haemoglobin, which is about 300 times more stable than the oxygen-haemoglobin complex. This prevents haemoglobin in the red blood corpuscles from carrying oxygen round the body and ultimately resulting in death.
New answer posted
11 months agoContributor-Level 10
Since, anhydrous HF is a covalent compound and weak acid due to high bond dissociation energy. AlF3does not dissolve in HF.
Whereas NaF is ionic compound.
3NaF + AlF3 → Na3 [AlF6]
Na3 [AlF6] + 3BF3 (g)→ AlF3 + 3Na+ [BF]–
New answer posted
11 months agoContributor-Level 10
B-Cl bond has dipole moment because of polarity. In BCl3, since the molecule is symmetrical (planar), the polarities cancel out and hence the dipole moment is zero
New answer posted
11 months agoContributor-Level 10
1.6. A mass percent of 69% means that 100 g of nitric acid solution contains 69 g of nitric acid by mass.
Molar mass of nitric acid HNO3= 1 + 14 + (3x16) = 63 gmol-1
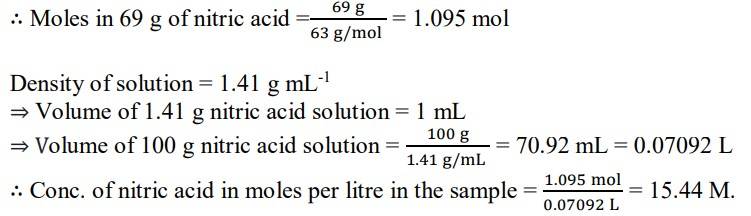
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers

