Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
Limestone:
- A raw material for cement.
- It is used as a building material in the form ofmarble and in the manufacture of quick lime.
- used in the manufactureof high quality paper. It is also used as anantacid, mildabrasive in tooth paste, aconstituent of chewing gum, and a filler incosmetics.
Cement:
- It is used in concrete and reinforcedconcrete, in plastering and in the construction
- of bridges, dams and buildings
Plaster of Paris:
- It is used in the building industry as well as plasters.
- It is usedfor immobilising the affected part of organ wherethere is a bone fracture or sprain.
- It is alsoemployed in dentistry, in ornamental work andfor making ca
New answer posted
11 months agoContributor-Level 10
Since group 1 hydroxides and carbonates due to large size contain higher hydration energy than the lattice energy so, they are easily soluble in water. Whereas, in magnesium and calcium due to small size their lattice energy dominates over hydration energy they are sparingly soluble in water.
New answer posted
11 months agoContributor-Level 10
BeCl2 (vapour)
In the vapour state, it exists as a chloro-bridged dimer.
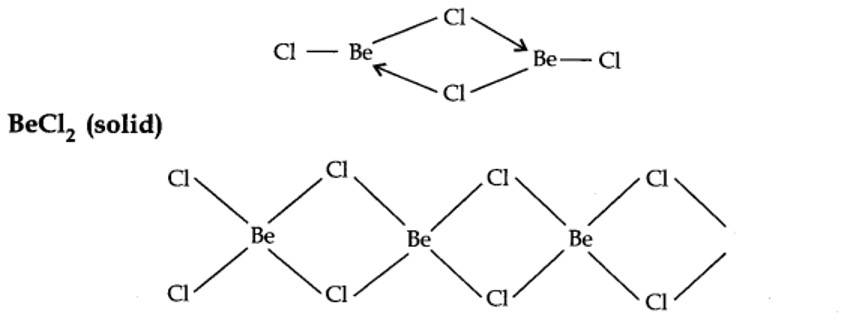
New answer posted
11 months agoContributor-Level 10
(i) Caustic soda
It is used in (a) the manufacture of soap, paper, artificial silk and a number of chemicals, (b) in petroleum refining, (c) in the purification of bauxite, (d) in the textile industries for mercerising cotton fabrics, (e) for the preparation of pure fats and oils, and (f) as a laboratory reagent.
(ii) Sodium carbonate
(a) It is used in water softening, launderingand cleaning.
(b) It is used in the manufacture of glass, soap, borax and caustic soda.
(c) It is used in paper, paints and textileindustries.
(d) It is an important laboratory reagent bothin qualitative and quantitative analysis
(iii) Quick lime
(a) It is an i
New answer posted
11 months agoContributor-Level 10
(i) Magnesium is burnt in air to form magnesium oxide and magnesium nitride.
2Mg + O2 → 2MgO
3Mg + N2 → Mg3N2.
(ii) Quick lime is heated with silica above 1273 K to obtain calcium silicate
CaO+SiO2 → CaSiO3.
(iii) Chlorine reacts with slaked lime to form calcium hypochlorite- a constituent of bleaching powder.
2Ca (OH)2 + 2Cl2 → CaCl2 + Ca (OCl)2 + 2H2O.
(iv) Calcium nitrate is heated to obtain CaO, NO2 and O2.
2Ca (NO3)2→2CaO+4NO2+O2.
New answer posted
11 months agoContributor-Level 10
(i) Sodium metal is manufactured by electrolysis of a fused mass of NaCl 40% and CaCl2 60% in Down's cell at 873 K, using iron as cathode and graphite as anode. Na is liberated at the cathode.
At cathode:
Na+ + e– → Na (l)
At anode:
2Cl– (melt) → Cl2 (g) + 2e–.
(ii) Sodium hydroxide is manufactured by electrolysis of an aqueous solution of NaCl (brine) in Castner-Kellner cell.
At cathode:
Na+ + e– → Na
2Na + Hg → Na – Hg + 2H2O
2Na- Hg + 2H2O → 2NaOH +H2 +Hg
At anode:
Cl– – e– → Cl
Cl + Cl→ Cl2
(iii) Sodium peroxide:
Sodium is heated in excess of oxygen to form sodi
New answer posted
11 months agoContributor-Level 10
(a) Nitrates of both group 1 and group 2 elements are soluble in water because hydration energy is more than the lattice energy.
Nitrates of both group 1 and group 2 elements are thermally unstable but they decompose differently except LiCO3 e.g.
2NaNO3 →2NaNO2 + O2
2KNO3 →2KNO2 + O2
4LiNO3 →2MgO + 4 NO2 + O2
2Mg (NO3)2 →2MgO + 4NO2 + O2
(b) Carbonates of group 1 elements are soluble in water except Li2CO3 They are also thermally stable except Li2CO3.
Li2CO3 →Li2O + CO2
Group 2 carbonates are insoluble in water because their lattice energy is higher than hydration energy.
Thermal stability of carbonates of group 2 inc
New answer posted
11 months agoContributor-Level 10
Li2CO3 is a covalent compound whereas Na2CO3 is an ionic compound. Therefore, lattice energy of Na2CO3 is higher than that of Li2CO3. Thus, LiCO3 is decomposed at a lower temperature as compared to Na2CO3.
New answer posted
11 months agoContributor-Level 10
Potassium carbonate cannot be prepared by Solvay process because potassium hydrogencarbonate is too soluble to be precipitated by the addition of ammonium hydrogencarbonate to a saturated solution of potassium chloride.
New answer posted
11 months agoContributor-Level 10
Solvay process is also known as ammonia soda process, wherein carbon dioxide is passed through a brine solution (containing about 28 % NaCl) which is saturated with ammonia to form sodium carbonate.
2NH3 + H2O + CO2 → (NH4)2CO3
(NH4)2CO3 + H2O + CO2 → 2NH4HCO3
NH4HCO3 + NaCl → NaHCO3↓ + NH4Cl
The precipitate of sodium bicarbonate is filtered, dried and ignited to form sodium carbonate.
2NaHCO3? →? Na2?CO3? + CO2 ?+ H2?O
The carbon dioxide required for the reaction can be obtained by heating limestone (calcium carbonate) to 1300 K in a lime klin. Lime dissolves in water to form calcium hydroxide which is then transferred to the ammo
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers
