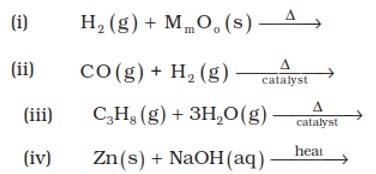Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
- Physical appearance: All the alkali metals are silvery white, soft and light metals.
- Density: Because of the large size, these elements have low density which increases down the group except for potassium which is lighter than sodium (most likely due to an unexpected increase in the atomic size.).
- The melting and boiling points of the alkali metals are low indicating weak metallic bonding due to the presence of only a single valence electron in them.
- Atomic volume:The atomic volume, atomic and ionic radii rise as the group number reduces from Li to Cs.
- Melting and boiling points:The weak crystal lattice bonding causes low melting and boili
New answer posted
11 months agoContributor-Level 10
Answer: (i) 3H2? (g)+2MoO3? ? Mo2? O3? +3H2? O (l)
(ii) CO (g) + H2 (g)? CH3OH
(iii)C3H8 (g) + 3H2O (g)? 3CO + 7H2 (g)
(iv) Zn (s) + NaOH (aq)? Na2ZnO2 (s) + H2 (g)
New answer posted
11 months agoContributor-Level 10
The differences between old and modern periodic table are as follows:
| Mendeleev's Table (Old Periodic Table) | Modern Periodic Table (Long Form Table) |
| Arrangement of elements are done on atomics weights (mass) | Arrangement of elements are done on atomic number |
| Contains 66 elements | Contains 118 known elements |
| Noble Gases not mentioned in old periodic table because it was not discovered | Noble gases are included in Group 18 |
| Transistion elements were placed with other elements | Transition elements are placed in seperate block |
New answer posted
11 months agoContributor-Level 10
NCERT Environmental Chemistry short questions and answers are provided in this artciles. These types of questions help in boosting the marks.
New answer posted
11 months agoContributor-Level 10
Yes, Class 11 Chemistry Environmental Chemistry notes are benefical in quick revision. Environmental Chemistry notes must consist of detailed explanation of topics, formulas, diagrams and reactions.
New answer posted
11 months agoContributor-Level 10
With the help of subject experts at Shiksha we have provided the NCERT solutions for environmental chmistry class 11 with explanation through this article.
New answer posted
11 months agoContributor-Level 10
Students can use the Class 11 Chemistry hydrocarbons shorts notes and question for quick resion and self assessment. After self assessment, it become easy to identify the weaker topics.
New answer posted
11 months agoContributor-Level 10
Yes, it is important to solve the Hydrocarbons ncert class 11 solved exercises for better understanding of topics. If the students faces any issue while solving the NCERT Class 11 Chemistry Hydrocarbons textbook exercises that means they must complete their conceptual knowledge.
New answer posted
11 months agoContributor-Level 10
Students can check the CBSE Class 11 Chemistry text book for NCERT solutions for class 11 hydrocarbons with diagrams. Students must practice the diagram solutions to master the questions.
New answer posted
11 months agoContributor-Level 10
As per NCERT the purification of an organic compounds are done in three ways - sublimation, crystallization, and distillation. The selection of the technique is based on the properties of compound and present of impurities.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers