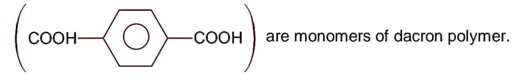Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoNew answer posted
7 months agoContributor-Level 9
2700 kJ energy released from 180 gm (1 mole) of glucose
1 kJ energy released from
10000 kJ energy released from
Amount of glucose =
New answer posted
7 months agoContributor-Level 10
Hydrogen peroxide reduces iodine to iodide ion is basic medium as;
New answer posted
7 months agoContributor-Level 10
w = 20 g
Mole of Na2O=
1 mole of Na2O gives 2 mole of NaOH
mole of Na2O gives moles of NaOH
Molarity of NaOH solution
= 1.29 M
=
Ans. = 13
New answer posted
7 months agoContributor-Level 9
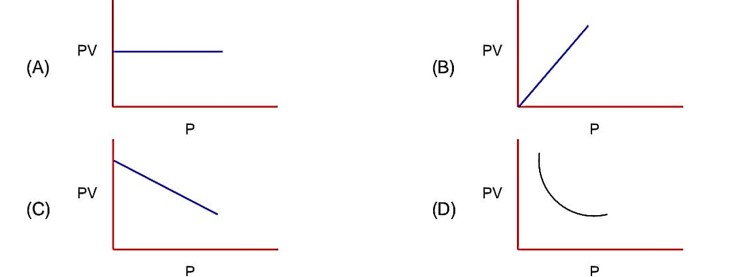
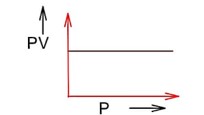
PV = nRT PV = constant (at constant T)
Pressure increases & volume decreases, PV remains constant at constant T.
New answer posted
7 months agoContributor-Level 9
B. O. D. value < 5 ppm for clean water and B.O.D value of polluted water
New answer posted
7 months agoContributor-Level 9
Portland cement contains
Dicalcium silicate = 26%
Tricalcium silicate = 51%
Tricalcium aluminate = 11%
Hence major percentage is of tricalcium silicate
New answer posted
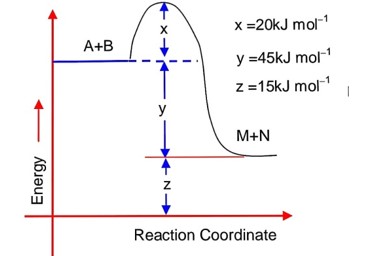
7 months agoContributor-Level 10
Applying :
Assuming the system attains a final temperature of T (Such that 300 < T < 60)
(Heat lost by N2 of container l) = (Heat gained by N2 of container II)
14 (300 – T) = T – 60
New answer posted
7 months agoTaking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers