Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
Lithium salts are extensively hydrated due to high hydration enthalpy of Li+
(order of polarizing power)
New answer posted
7 months agoContributor-Level 10
Here, number of unpaired electrons, n = 1
Spin only moment ;
= 173 * 10-2 B.M
New answer posted
7 months agoContributor-Level 10
Ammonium salt in rain drop resulting wet deposition
Oxides of N & S settle down on ground as dry deposition (SO2).
New answer posted
7 months agoContributor-Level 10
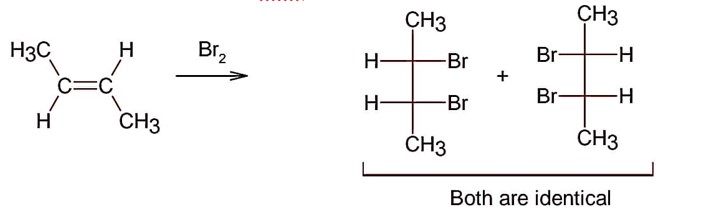
Electrophilic addition of bromine to an alkene is anti-addition, in which cis-alkene gives two enantiomers and trans – alkene gives meso form
Here; trans-but-2-ene will give meso products
New answer posted
7 months agoContributor-Level 10
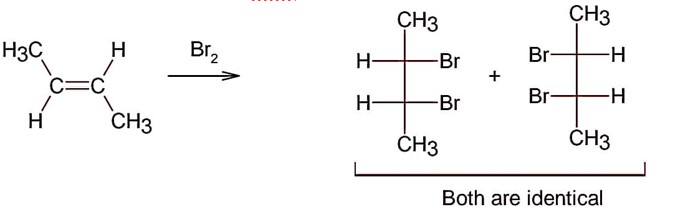
Electrophilic addition of bromine to an alkene is anti-addition, in which cis-alkene gives two enantiomers and trans – alkene gives meso form
Here; trans-but-2-ene will give meso products
New answer posted
7 months agoContributor-Level 10
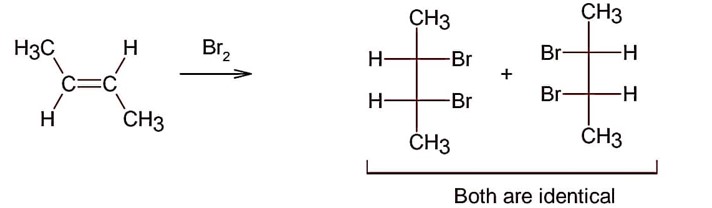
Electrophilic addition of bromine to an alkene is anti-addition, in which cis-alkene gives two enantiomers and trans – alkene gives meso form
Here; trans-but-2-ene will give meso products
New answer posted
7 months agoContributor-Level 10
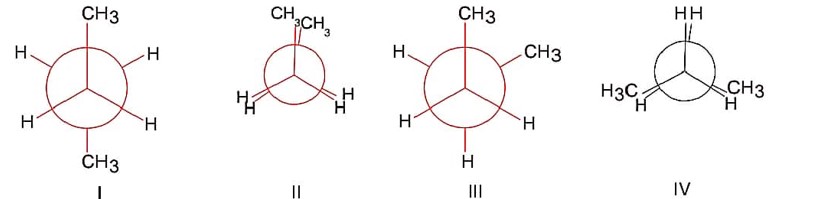
Structure (I) is anti conformer.
Structure (II) is fully eclipsed conformer.
Structure (III) is skew or gauche conformer.
Structure (IV) is partially eclipsed.
order of stability
(I) > (III) > (IV) > (II)
Order of P.E. is (II) > (IV) > (III) > (I).
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers