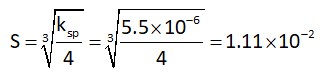Ncert Solutions Chemistry Class 11th
Get insights from 2k questions on Ncert Solutions Chemistry Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
Bond dissociation enthalpy is the energy required to break 1 mole of bond according to experimental data
Bond energy : Cl2 > Br2 > F2 > l2
New answer posted
7 months agoContributor-Level 10
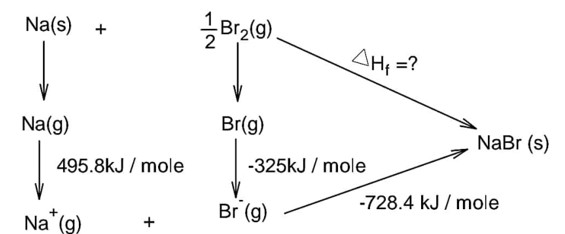
Bond dissociation energy of Br2 = 228 kJ/mole
Note : In question is neglect
New answer posted
7 months agoContributor-Level 10
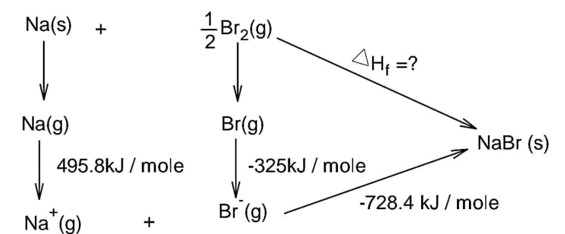
Bond dissociation energy of Br2 = 228 kJ/mole
Note : In question is neglected
New answer posted
7 months agoContributor-Level 10
NH2CN (s) + O2 (g) ® N2 (g) + O2 (g) + H2O (l)
=
So, magnitude of
New answer posted
7 months agoContributor-Level 10
(A) Isothermal process Temperature is constant throughout the process
(B) Isochoric process Volume is constant throughout the process
(C) Isobaric process Pressure is constant throughout the process
(D) Adiabatic process No exchange of heat between system and surrounding
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers