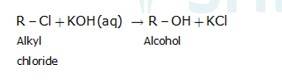Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
7.41
Copper metal on reaction with HNO3 gets oxidized and give different by-products depending on the temperature, concentration of the acid and the copper metal undergoing oxidation.
The reaction of copper with concentrated and dilute HNO3 is as shown below:
3Cu + 8HNO3 (did.) 3Cu (NO3)2 + 2NO +4H2O
Cu + 4HNO3 (conc.) Cu (NO3)2 + 2NO2 +2H2O
New answer posted
a year agoContributor-Level 10
This is a classic question from the chapter Haloalkanes and Haloarenes. Let us solve each one as follows:
(1) When n - butyl chloride is treated with alcoholic KOH, the formation of but - l - ene takes place. This reaction is a dehydrohalogenation reaction.
When N-butyl chloride or 1-chlorobutane is treated with alcoholic potassium hydroxide (KOH), elimination reaction takes place. This leads to alkene formation. 1-butene is the major product here.
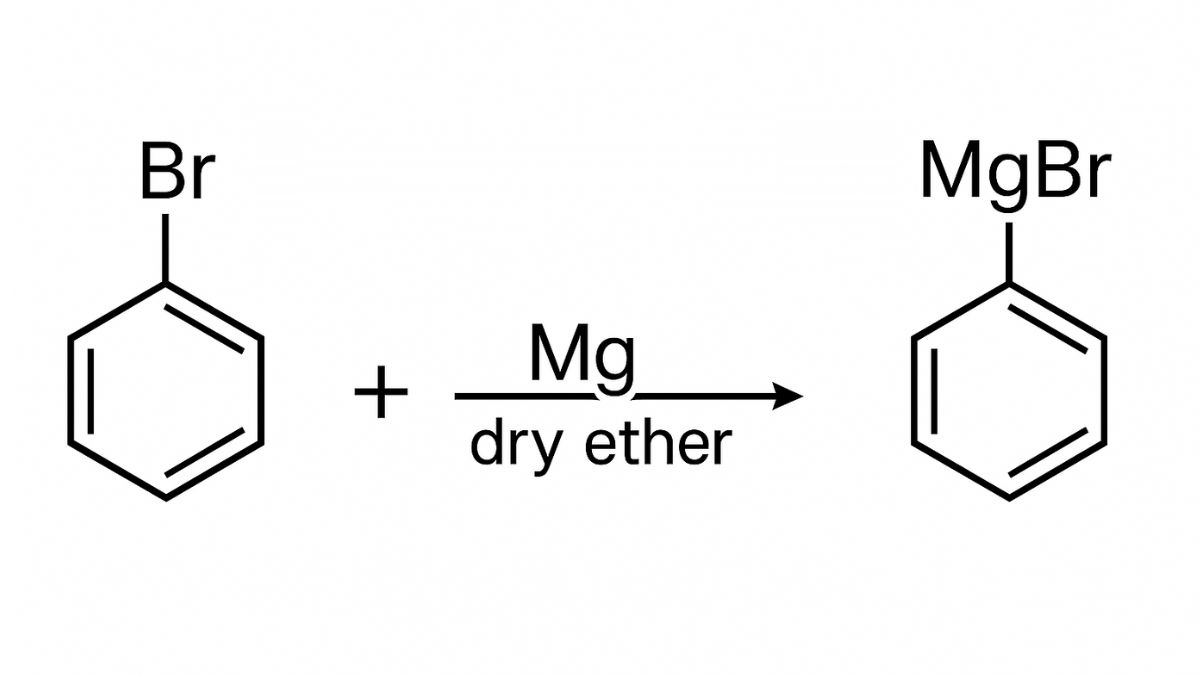
(ii) When Bromobenzene is treated with Mg in the presence of dry ether, it undergoes a reaction to produce phenylmagnesium bromide which is the Grignard reagent. This reagent is very re
New answer posted
a year agoContributor-Level 10
7.40
Ammonia is manufactured industrially by Haber's process. Nitrogen from gas is combined with hydrogen derived from natural gas (methane) in the ratio 1:3 giving rise to ammonia. The reaction is reversible and exothermic.

The optimum conditions for the manufacturing of ammonia are pressure 200*105 Pa, the temperature of 4700 K and iron oxide catalyst with a small amount of Al2O3 and K2O.
New answer posted
a year agoContributor-Level 10
7.39
Nitrogen is prepared in the laboratory by heating aqueous ammonium chloride (NH4Cl) with sodium nitrite (NaNO2) to form ammonium nitrite (NH4NO2), which is unstable. Ammonium nitrite breaks down to form nitrogen and water.
NH4Cl (aq) + NaNO2 (aq) - NH4NO2 + NaCl (aq)
NH4NO2 - N2 (g)+ H2O (l)
Small amounts of NO and HNO3 are also produced which can be removed by passing nitrogen gas through aqueous sulphuric acid containing potassium dichromate.
New answer posted
a year agoContributor-Level 10
There are two primary alkyl halides having the formulaC4H9Br, They are n - butyl bromide and isobutyl bromide.
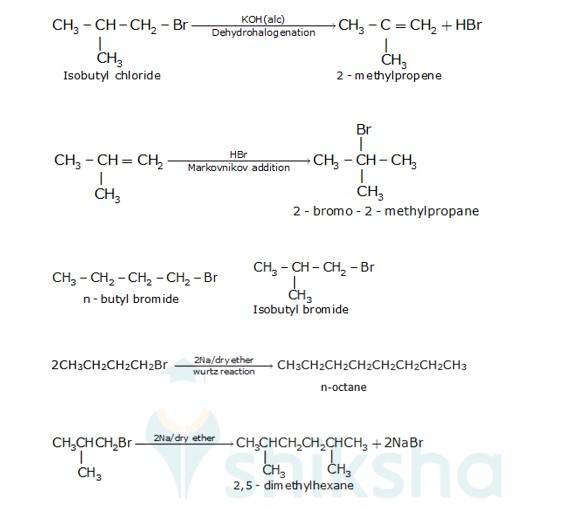
Therefore, compound (a) is either n-butyl bromide or isobutyl bromide.
Now, compound (a) reacts with Na metal to give compound (b) of molecular formula, C18H18 which is different from the compound formed when n-butyl bromide reacts with Na metal.
Hence, compound (a) must be isobutyl bromide. Thus, compound (d) is 2, 5-dimethylhexane.
It is given that compound (a) reacts with alcoholic KOH to give compound (b). Hence, compound (b) is 2- methylpropene.
Also, compound (b) reacts with HBr to give compound (c) which is an isomer of (a)
New answer posted
a year agoContributor-Level 10
An aqueous solution, KOH almost completely ionizes to give OH-ions. OH- ion is a strong nucleophile (see the imp. note below), which leads the alkyl chloride to undergo a nucleophilic substitution reaction to form alcohol.
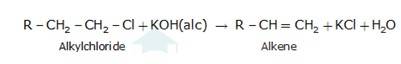
On the other hand, an alcoholic solution of KOH contains alkoxide (RO-) ion, it is a strong base. Thus, it can abstract a hydrogen ion from the beta-carbon of the alkyl chloride and form an alkene by eliminating a molecule of HCl.OH- ion is a much weaker base than RO- ion. Also, OH- ion is highly solvated (because more energy is released on solvation)in an aqueous solution and as a result, the basic character of OH-io
New answer posted
a year agoContributor-Level 10
7.38
The electronegativity of N (3.0) is higher than that of P (2.1). Due to this N-H bond is more polar than a
P-H bond. Also, P and H have the same electronegativity of 2.1 i.e., the P-H bond is non-polar. Therefore, PH3 does not form hydrogen form.
The structure of NH3 and PH3 with their electronegativity is represented below.

New answer posted
a year agoContributor-Level 10
(i) Propene to propan-1-ol
It is an antimarkovnikoff reaction in which under the presence of a peroxide an alkene undergoes substitution, wherein the halo group is attached to that carbon which has least no. Of alkyl groups attached to it.
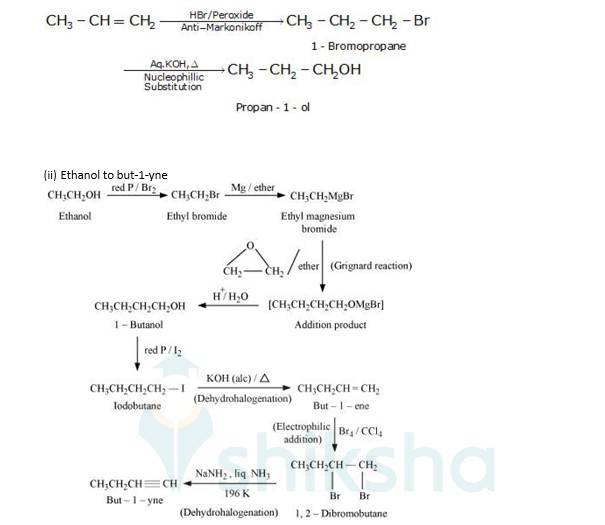
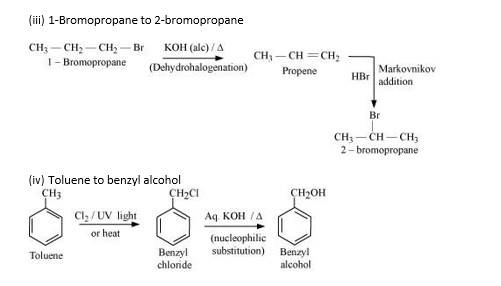
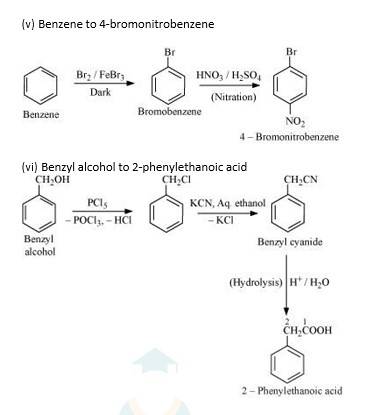
New answer posted
a year agoContributor-Level 10
7.37
Chemical reactivity of group 15 elements towards hydrogen, oxygen, halogens, and metals are discussed below.
Reactivity towards hydrogen: Group 15 elements react with H to form hydrides of type EH3 where E=N, P, As, Sb or On moving down from NH3 to BiH3, the stability of the hydrides decreases. For example, the P-H bond in PH3 is less stable than the N-H bond in NH3. The strength of the E-H bond gets weaker as the size of the central atom increases.
Stability order of E-H bond (where E is group 15 elements) can be represented as
N-H > P-H > As-H >Sb-H > Bi-H
Reactivity towards oxygen: Group 15 elements react with O to form oxides
New answer posted
a year agoContributor-Level 10
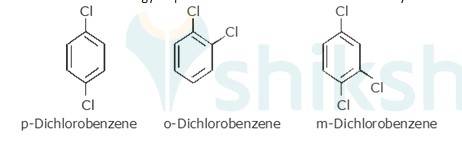
p-Dichlorobenzene is more symmetrical than o-and m-isomers. Because it fits more closely and easily in the crystal lattice than o-and m-isomers. Therefore, more energy is required to break the crystal lattice of p-dichlorobenzene. Therefore, p-dichlorobenzene is symmetrical and has a higher melting point and lower solubility than o-and m-isomers due to strong force of attraction in crystal.
Therefore more energy required to break lattice and will not easily be soluble.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers