Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
Hydrolysis by KOH results in the formation of the carbocation. Those compounds which leads to the formation of stable carbocation are easily hydrolysed.C6H5CH2Cl leads to formation of 1°- carbocation, while C6H5CHClC6H5 forms 2°-carbocation, which is more stable than 1°-carbocation. Hence C6H5CHClC6H5, is hydrolyzed more easily than C6H5CH2Cl by aqueous KOH.
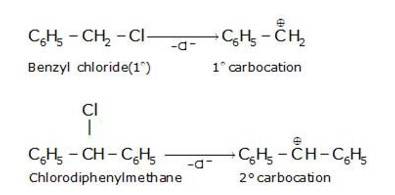
New answer posted
a year agoContributor-Level 10
The reaction involves the approaching of the nucleophile to the carbon atom to which the leaving group is attached. When the nucleophile is sterically hindered (does not have any free space or very crowded), then the reactivity towards SN2 displacement decreases. Due to the presence of substituents, hindrance to the approaching nucleophile increases in the following order.
- Bromopentane < 2-bromopentane < 2-Bromo-2-methylbutane. The structures are shown below:
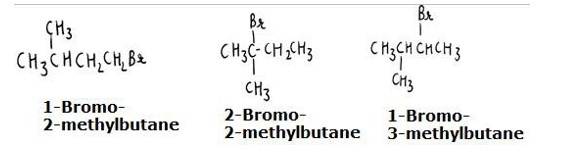
Hence, the increasing order of reactivity towards SN2 displacement is:
- Bromo-2-methylbutane < 2-Bromopentane < 1-Bromopentane
- The stearic hinderance in alkyl halides increases in the order of 1° < 2 < 3, the increasing order of reactivity towards SN2 displacement is given as-
3° < 2 < 1.
The structures are given below:

Hence, the given set of compounds can be arranged in the in
New answer posted
a year agoContributor-Level 10
7.2
Nitrogen atom can bond with another nitrogen atom by strong p–p overlap resulting in NN. The triple bond in N2 has high bond strength resulting in high bond dissociation energy. Phosphorous do not show this property of p–p overlap. Hence, nitrogen is less reactive than phosphorous.

New answer posted
a year agoContributor-Level 10
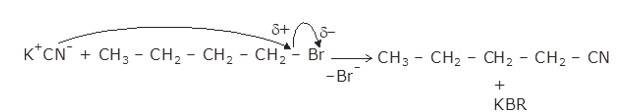
The given reaction is a nucleophillic reaction:

The given reaction is an SN2 reaction. In this reaction, CN acts as the stronger nucleophile and attacks the carbon atom to which Br is attached in nBuBr.
And, CN- ion is an ambident nucleophile and can attack through both C and N. In this case, it attacks through the C-atom.
The mechanism is shown below:
New answer posted
a year agoContributor-Level 10
7.1
The general characteristics of Group 15 elements are:
Electronic configuration: All Group 15 elements have 5 electrons in their valence The general electronic configuration of these elements is ns2 np3
Oxidation state: Group 15 elements have 5 valence electrons and they require 3 more electrons to complete their However, the gaining of 3 electrons is difficult
Atomic size: Atomic size increases as we move down the group due to increase in the number of
Ionisation enthalpy: Ionisation enthalpy decreases as we move down the group because of increase in atomic
Electronegativity: Electronegativity decreases on moving down the group due to in
New answer posted
a year agoContributor-Level 10
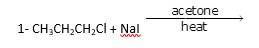
It is a simple substitution reaction with Cl being replaced by iodide ion.
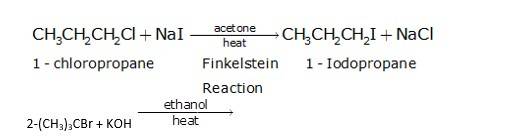
Since ethanol is a alcohol, so in presence of alcoholic KOH, alkyl chloride undergo elimination reaction, which results in the removal of proton and being substituted by a bromide ion.
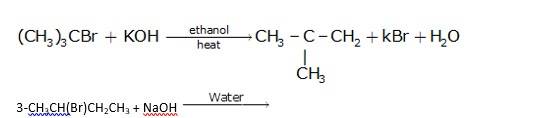
It is also a simple substitution reaction in which under the presence of aqueous NaOH, bromide ion is replaced by hydroxide ion as it is a better leaving group than hydroxide ion.
It is a simple substitution reaction in which a better leaving group leaves and is being substituted by an ion.
CH3CH2Br + KCN - CH3CH2CN + kBr
5-C6H5ONa + C2H5Cl
It is a simple substitution reaction in
New answer posted
a year agoContributor-Level 10
7.34
It is difficult to study the chemistry of radon because it is a radioactive substance having a half-life (the time period to decompose the substance half to its initial concentration) of only 3.82 days.
Radon belongs to the 18 group elements with chemical formula as Rn. In general, the elements at the bottom o periodic table are radioactive, they are very dangerous to study as they emit harmful radiations.
Also, compounds of radon such as RnF2 have not been isolated, they are still in the phase of discovery. They have only been identified by radiotracer technique and no any further properties have been determined.
New answer posted
a year agoContributor-Level 10
(i) Freon 12:- the chlorofluorocarbon compounds of methane and ethane are collectively known as freons. They are extremely stable, unreactive, non-toxic and non-corrosive. Example of Freon 12 is CCl2F2. It is mainly used in refrigeration and eventually makes its way into the atmosphere where it diffuses unchanged into the stratosphere. In stratosphere, Freon is able to initiate the radical chain reactions that can upset the natural ozone balance.
- DDT: - it stands for p, p'-Dichlorodiphenyltrichloroethane (DDT). It is mainly used as an
- Iodoform: - it was earlier used as an antiseptic but the antiseptic properties are due the liberation o
New answer posted
a year agoContributor-Level 10
Let us explain each question from Haloalkane and Haloarenes chapter one by one. Students who are currently in school and plan to take the CBSE board exam for class 12th soon, need to prepare all these questions. Let us get started.
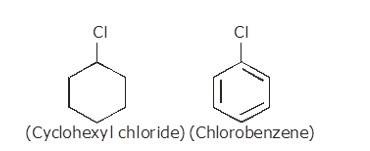
(i) The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
Sp2 hybrid carbon (s-character=33.33%) in chlorobenzene is more electronegative than a sp3-hybrid carbon (s-character=25%) in cyclohexylchloride, due to greater s-character. Thus, Carbon atom of chlorobenzene has less tendency to release electrons to Cl than carbon atom of cyclohexylchloride.
Chlorine atom in chlorobenzene is atta
New answer posted
a year agoContributor-Level 10
7.33
XeF6 + H2O XeO2F2 + HF
Balanced equation: XeF6 + 2H2O XeO2F2 +4HF. The steps for balancing the reaction are as follows:
1. The main element is First, check if the atoms of Xe on both sides are balanced. YES, they are.
2. Then take another element i.e. F. It is not balanced on the right side, there are 3 atoms of F missing. So, to balance it multiply HF by 4.
3. Now check the other secondary atoms which are oxygen and hydrogen. Oxygen is not balanced on the left side as there are 2 O atoms so multiply H2O by
4. Now check for the hydrogen They are balanced on both sides.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers