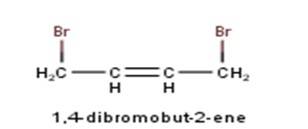
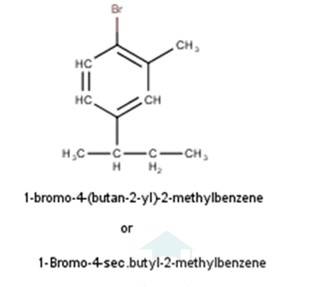
Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
In the presence of sulphuric acid (H2SO4), KI produces HI as follows : -
2KI + H2SO4 → 2KHSO4 + 2KI
Since H2SO4 is an oxidising agent, it oxidises HI (produced in the reaction to I2)
2HI + H2SO4 → I2 + SO2 +;
As a result, the reaction between an alcohol and HI to produce alkyl iodide cannot occur. Therefore, sulphuric acid is not used during the reaction of alcohols with KI. Instead, a non-oxidising acid such as H3PO4 is used in the reaction to get the desired product.
A few things you can remember here, while solving NCERT Solutions for Haloalkanes And Haloarenes.
Sulphuric acid is a powerful oxidising agent. We know this because the su
New answer posted
a year agoContributor-Level 10
7.31
In interhalogen compound ICl and I2 the atoms are bonded by covalent bonds
Reactivity refers to the rate at which a chemical species will undergo reaction in time. For reaction to take place the compound needs to be broken into separate elements first, then the individual elements react with other elements to form new compounds. So any compound which can easily break into their individual elements can react faster.
The covalent bonds between dissimilar atoms I and Cl atoms in ICl are weaker than between similar atoms in I2. Therefore the bond between ICL will break easily so the I and Cl atom will be easily available to form another
New answer posted
a year agoContributor-Level 10
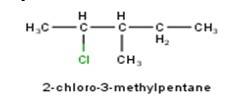
10.1 From the name of the compound it is clear that the parent ring is pentane and chloro and methyl groups are attached in the straight chain at 2nd and 5th position respectively. Hence, the structure is as follows:
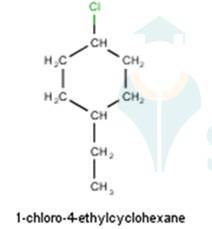
From the name of the compound given it is clear that the parent group is hexane with 2 attachments namely chloro and ethyl groups at 1 and 4 positions respectively. Hence, the structure is as follows: -
From the name of the compound given it is clear that the parent group is heptane with tertiary butyl and iodine groups attached at 4 and 3 positions respectively. Hence, the structure is as follows: -
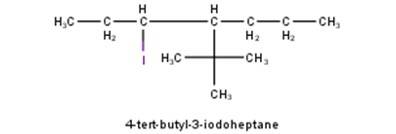
From the name of the comp
New answer posted
a year agoContributor-Level 10
8.48 To calculate magnetic moment of the complex species, we use the spin formula:
μ =√n(n+2) BM
When n= 1 | ⇒ μ = √1(1+2) ⇒ u= √3⇒ u=1.73 BM |
When n=2 | ⇒ μ = √2(2+2) ⇒ μ = √8 ⇒ μ = 2.83 BM |
When n= 3 | ⇒ μ = √3(3+2) ⇒ μ = 15 ⇒ μ = 3.87 BM |
When n = 4 | ⇒ μ = √ 4(4+2) ⇒ μ = √24 ⇒ μ = 4.899 BM |
When n= 5 | ⇒ μ = √5(5+2) ⇒ μ = √35 ⇒ μ = 5.92 BM |
1. [K4 [Mn(CN)6]
⇒μ = 2.2 BM (given)

We can see from the above calculation that the given value(2.2) is close to n=1. It means that it has only one unpaired electron Also in this complex Mn is in +2 oxidation state,i.e., as Mn2+.Thus when CN- ligands approach Mn2+ ion, The electrons in 3d do not pair up.
The atomic number of Manganese (Mn) is Z = 25
The electronic configuration of 25Mn= [Ar] 3d5 4s2
And, the electronic configuration of Mn2+=[Ar] 3d5
Thus CN- is a strong ligand.
The hybridization involved is d2sp3 forming inner orbital oc
New question posted
a year agoNew answer posted
a year agoContributor-Level 10
8.47 The given statement is true as explained below:
1. Atomic radii of the heavier transition elements (4d and 5d series) are larger than those of the corresponding elements of the first transition series through those of 4d and 5d series are very close to each (Lanthanoid contraction)
2. Due to stronger intermetallic bonding (M-M bonding), the melting and boiling points of heavier transition elements are greater than those of the first transition series
3. The ionization enthalpies of 5d series are higher than the corresponding elements of 3d and 4d
4. The heavier transition elements form low spin complexes whereas the elements of t
New answer posted
a year agoContributor-Level 10
8.47 The given statement is true as explained below:
1. Atomic radii of the heavier transition elements (4d and 5d series) are larger than those of the corresponding elements of the first transition series through those of 4d and 5d series are very close to each (Lanthanoid contraction)
2. Due to stronger intermetallic bonding (M-M bonding), the melting and boiling points of heavier transition elements are greater than those of the first transition series
3. The ionization enthalpies of 5d series are higher than the corresponding elements of 3d and 4d
4. The heavier transition elements form low spin complexes whereas the elements of t
New answer posted
a year agoContributor-Level 10
8.46
S.no | Ion | Configuration | Number of 3d electrons | No. of unpaired Electrons | 3d orbitals |
1 | Ti2+ | 3d2 | 2 | 2 | t22g e0g |
2 | V2+ | 3d3 | 3 | 3 | t32g e0 g |
3 | Cr3+ | 3d3 | 3 | 3 | t32g e0 g |
4 | Mn2+ | 3d5 | 5 | 5 | t32g e2 g |
5 | Fe2+ | 3d6 | 6 | 4 | t42g e2 g |
6 | Fe3+ | 3d5 | 5 | 5 | t32g e2 g |
7 | Co2+ | 3d7 | 7 | 3 | t52g e2 g |
8 | Ni2+ | 3d8 | 8 | 2 | t62g e2 g |
9 | Cu2+ | 3d9 | 9 | 1 | t62g e3g
|
Note: In an octahedral field, the d-orbitals split into two sets of orbitals, the set of orbitals ( dxy, dyz, dxz) with lower energy is called t2g and the set of orbitals (dx2-y2 and dz2) with higher energy is called eg
New answer posted
a year agoContributor-Level 10
8.45 (i) Electronic configuration:
In the first transition series, 3d orbitals are progressively filled while in the second and third transition series, 4d and 5d orbitals are filled. However the first series shows only two exceptions Cr and Cu, both have a single electron in the 4s orbital ( 3d5 4s1, 3d10 4s1) but the second series shows more exceptions. Similarly, third series elements show exceptions. Thus in the same vertical column, in a number of series, the electronic configuration of the three series are not similar at all.
(ii) Oxidation states:
The number of oxidation states shown by the elements in the middle of each ser
New answer posted
a year agoContributor-Level 10
8.44 Promethium (Pm) has atomic number 61. Hence electronic configuration of Promethium is [Xe]544f5 5d0 6s2
Protactium (Pa) has atomic number 91. Hence electronic configuration of Protactium is [Rn]86 5f2 6d1 7s2
Mendelevium (Md) has atomic number 101. Hence electronic configuration of Mendelevium is [Rn]86 5f14 6d0 7s2
Meitnerium (Mt) has atomic number 109. Hence electronic configuration of Meitnerium is [Rn]86 5f14 6d7 7s2
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers