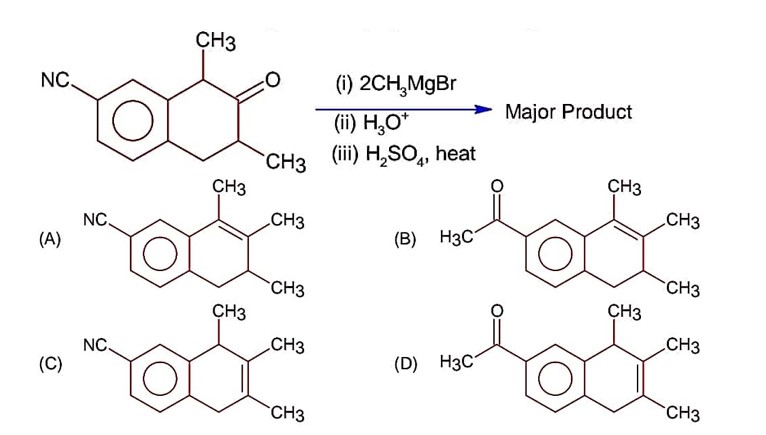
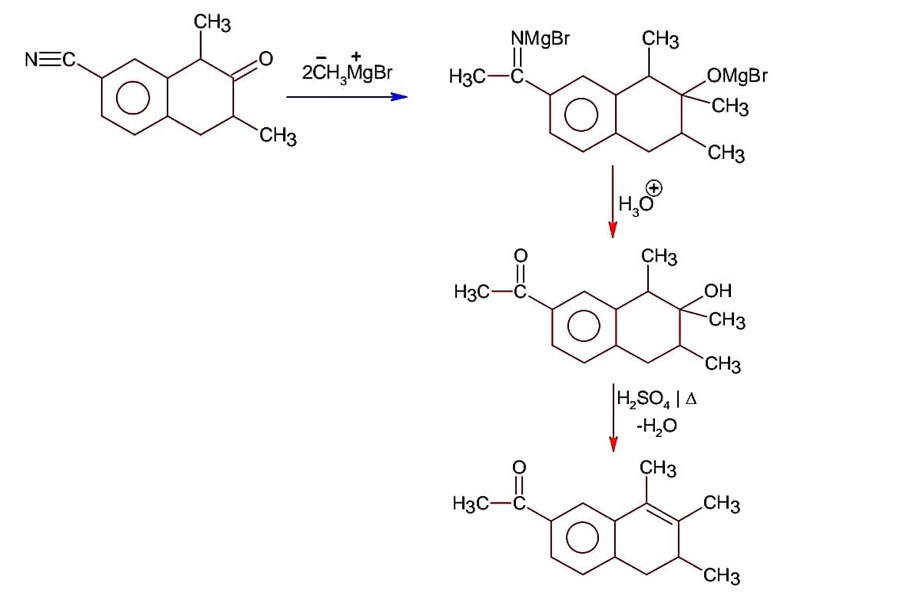
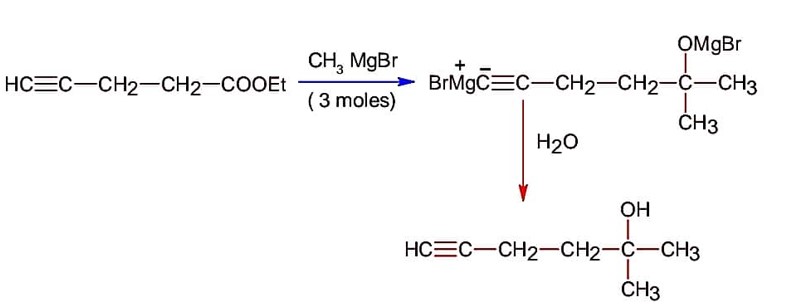
Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 10
O2F2 oxidises plutonium to PuF6 and the reaction is used in removing plutonium as PuF6 from spent nuclear fuel.
New answer posted
9 months agoContributor-Level 10
When red phosphorus is heated in a sealed tube at 803 K, a - black phosphorus is formed.
New answer posted
9 months agoContributor-Level 10
Let molarity of KMnO4 = x
n = 5 n = 1 Ferric sulphate
equivalent of KMnO4 = equivalent of FeSO4
5 * x * 10 = 1 * 0.1 * 10
x = 0.02 M
Strength = (0.02 * 158) = 3.16g/L
= 316 * 10-2 g/L
Ans. = 316
New answer posted
9 months agoContributor-Level 10
Dumas method,
Moles of N in N, N-dimethylaminopentane (C7H17N)
= 11.25 mol
= 1125 * 10-2 mol
Ans. = 1125
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers