Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
9 months agoContributor-Level 9
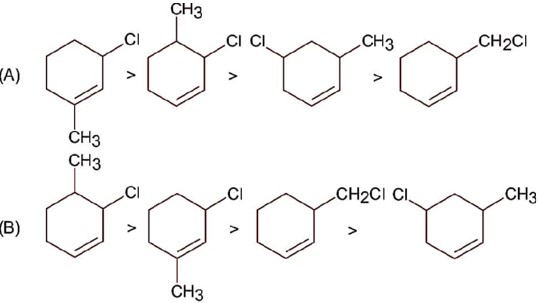
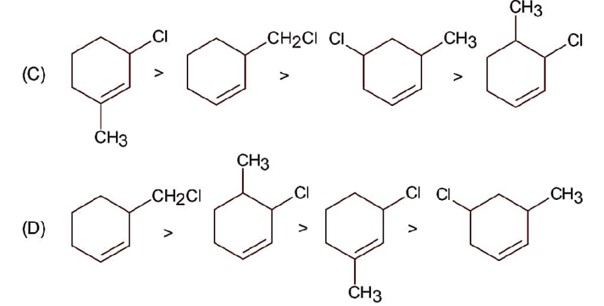
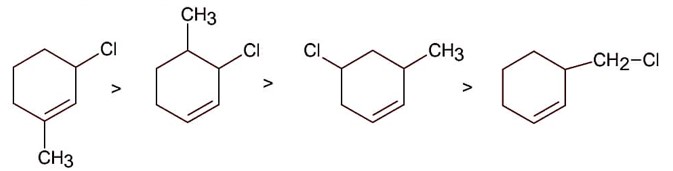
Compound react with SN1 mechanism in presence of polar protic solvent, which follows carbocation forming path.
Hence correct order is.
New answer posted
9 months agoContributor-Level 9
In electrolytic reduction of Al2O3, cryolite (Na3AlF6) is used to increase conductivity & decrease melting point. Oxidation state of Al in cryolite (Na3AlF6) is (+3).
New answer posted
9 months agoContributor-Level 9
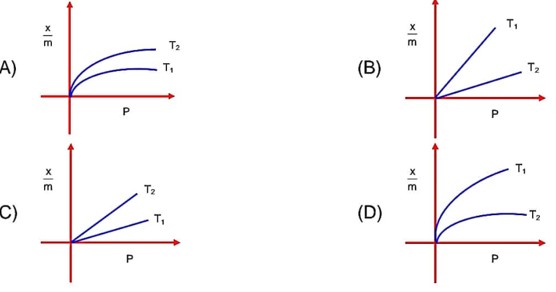
By increases in temperature absorption decreases, T1 > T2 means higher absorption at T2 temperature.
New answer posted
9 months agoContributor-Level 9
HgS, PbS, CuS, Sb2S3, As2S3, & CdS are given sulphides
CdS, PbS, As2S3 & CuS are soluble in 50% HNO3 but Sb2S3 & HgS are not soluble.
Ans. = 4
New answer posted
9 months agoContributor-Level 10
Applying :
Assuming the system attains a final temperature of T (Such that 300 < T < 60)
(Heat lost by N2 of container l) = (Heat gained by N2 of container II)
14 (300 – T) = T – 60
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers