Chemistry NCERT Exemplar Solutions Class 11th Chapter Nine
Get insights from 89 questions on Chemistry NCERT Exemplar Solutions Class 11th Chapter Nine, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry NCERT Exemplar Solutions Class 11th Chapter Nine
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
(i) PBS (s)+4H2O2 (aq) → PbSO4 (s)+4H2O (l)
(ii) CO (g)+2H2 (g) → CH3OH (l)
New answer posted
11 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Ice has a highly ordered 3D hydrogen bonded structure. Each oxygen atom is surrounded tetrahedrally by four other oxygen atoms at a distance of 276 pm.
New answer posted
11 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
NaH belongs to Saline hydrides and B2H6 and H2O belong to molecular hydrides.
New answer posted
11 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
These are formed by many d-block and/block elements. However, the metals of group 7, 8 and 9 do not form hydride. Unlike saline hydrides, they are almost always non- stoichiometric, being deficient in hydrogen. For example, LaH 2.87, YbH 2.55, TiH 1.5-1.8, ZrH 1.3-1.75. Dihydrogen forms molecular compounds with most of the jp-block elements.
Most familiar examples are CH4, NH3, H2O and HF.
New answer posted
11 months agoContributor-Level 10
This is a short answer type question as classified in NCERT Exemplar
Water-gas shift reaction: The water-gas shift reaction is a chemical reaction in which carbon monoxide reacts with steam in the presence of a catalyst to form carbon dioxide and hydrogen.
CO+H2+H2O → CO2+2H2
New answer posted
11 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
Sodium reacts with Dihydrogen to form sodium hydride which is a crystalline ionic solid.
2Na + H2 → 2Na + H-
It reacts with H2O to produce H2 gas
2NaH+2H2O → 2NaOH+2H2
Although Na+H- does not conduct electricity in the solid state, the electrolysis of its melt produces H2 at the anode and Na at the cathode.
At cathode At anode
Na + H- (l) → &nbs
New answer posted
11 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
It is lithium hydride (LiH) because it has significant covalent character due to the smallest alkali metal, LiH is very stable. It is almost nonreactive towards oxygen and chlorine. It reacts with Al2Cl2 to form lithium aluminium hydride.
8LiH+ Al2Cl6 → 2LiAlH4 → LiCl
New answer posted
11 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
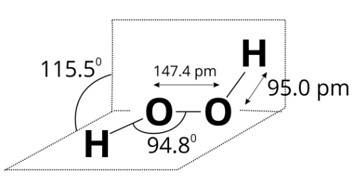
Since, a colourless liquid 'A' contains only hydrogen and oxygen and decomposes slowly on exposure to light but is stabilised by addition of urea, therefore, liquid A may be hydrogen peroxide. A is H2O2.
Structure of H2O2 is given below.
New answer posted
11 months agoContributor-Level 10
This is a long answer type question as classified in NCERT Exemplar
Mass of H2O2 = 68 g.
Mol. Mass of H2O2=34gmol-1
∴1L of M solution of H2O2 will contain H2O2=34*5 g
∴2L of 5 M solution will contain H2O2=34*5*2=340g
or 200 mL of 5 solution will contain H2O2 = 200 = 34 g
Now 2H2O2 → 2H2O+O2
64 g 32g
Now 68 g of H2O2 on decomposition will give O2=32g
∴34g of H2O2 on decomposition will give O2 = 34 =16 g
New answer posted
11 months agoContributor-Level 10
(i) Industrial preparation: H2O2 is prepared by the auto-oxidation of 2- alkylanthraquinols
2-ethylanthraquinol ↔ H2O2+ oxidised product
2Fe2+ (aq)+2H+ (aq)+H2O2 → Fe3 (aq)+2H2O (l)
PbS (s)+4H2O2 (l) → PbSO4 (s)+4H2O (l)
(ii) Reducing action of hydrogen peroxide
2MnO4-+6H++5H2O2→ 2Mn2++8H2O+5H2
HOCl+H2O2 → H3O++Cl-+O2
Oxidising action of hydrogen peroxide
2Fe2++H2O2→ 2Fe3++2OH-
Mn2++H2O2 → Mn4++2OH-
Acidic properties of H2O2
I2+H2O2+2OH-→ 2I-+2H2O+H2
2MnO4-+3H2O2→ 2MnO2+3O2+2H2O+2OH-
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers
