Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
6 months agoContributor-Level 10
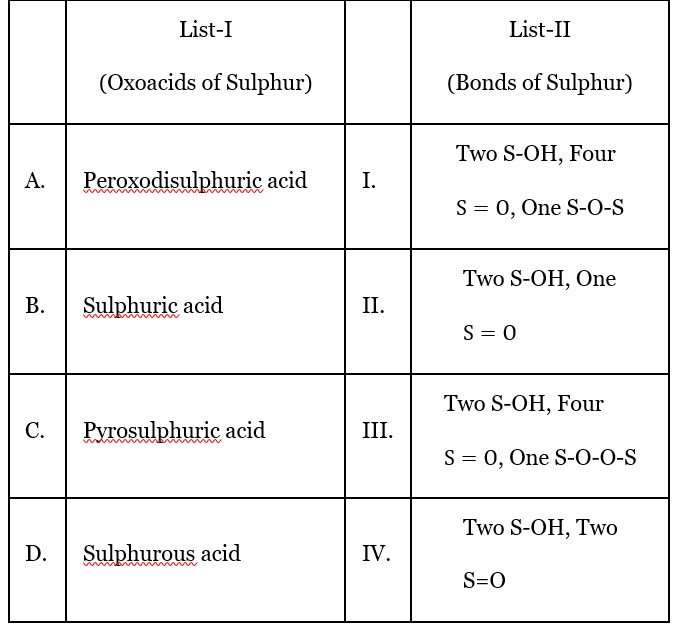
H? S? O? , peroxodisulphuric acid (Two S-OH, Four S = O, One S – O – O – S )
H? SO? , sulphurous acid (Two S-OH, One S = O bond)
H? S? O? , pyrosulphuric acid (Two S-OH, Four S=O, One S-O-S bond)
H? SO? , sulphuric acid (Two S – OH, Two S = O )
New answer posted
6 months agoContributor-Level 10
Fe? O? + CO → 2FeO + CO?
The above reaction takes place at 500 – 800 K in blast furnace.
New answer posted
6 months agoContributor-Level 10
Pumice stone is an example of solid sol. In this type of colloid, the dispersion medium is solid and the dispersion phase is gas.
New answer posted
6 months agoContributor-Level 10
One edge of an cube is common to four unit cells. Hence One edge centre OV contribute 1/4 to one unit cell.
New answer posted
6 months agoContributor-Level 10
Eutrophication occurs when the water body becomes overly enriched with nutrients. Eutrophication leads to decrease in the level of dissolved oxygen (DO) in water bodies.
New answer posted
6 months agoContributor-Level 10
Kolbe's electrolysis is a process where an aqueous solution of sodium and potassium salt undergoes electrolysis to lead to the formation of a symmetrical alkene. Principle of this technique is that at anode, the carboxylate ion undergoes oxidation and loses carbon dioxide (CO? ). Similarly, at cathode the water decomposes to form hydrogen gas. The alkyl radicals formed in these two cases combine to form alkenes.
New answer posted
6 months agoContributor-Level 10
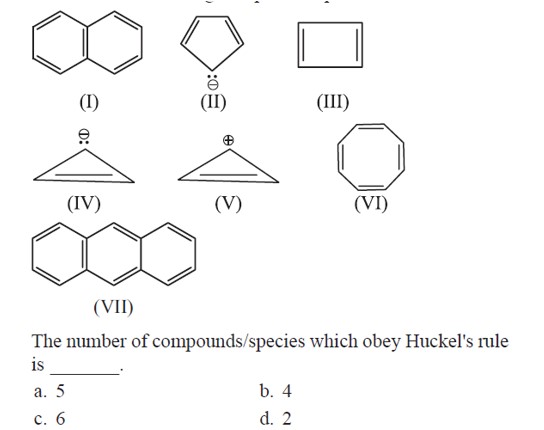
Acc. to Huckel rule, A compond/molecule is said to be aromatic if it have cyclic conjugate and [4n + 2]π electron system
⇒ Molecule must be sp² hybridised
⇒ Molecule must be planar.
New answer posted
6 months agoContributor-Level 10
Reduction half reaction.
Cr? O? ²? + 14H? + 6e? → 2Cr? ³ + 7H? O
Oxidation half reaction
SO? ²? + H? O → SO? ²? + 2e? ] * 3
Oxygen is balanced by adding water and hydrogen is balanced by adding H? and the charge is balanced by electrons.
Add ( eq. (i) + (3 * eq. (ii) )
Cr? O? ²? + 3SO? ²? + 8H? → 2Cr? ³ + 3SO? ²? + 4H? O
a = 1 b = 3 c = 8
New answer posted
6 months agoContributor-Level 10
Keq. = ( [C] [D])/ ( [A] [B])
Keq = (10 * 6)/ (2 * 3) = 10
ΔG° = −2.303RTlog Keq. = −2.303 (2) (300) (log 10)
= −1381.8cal (? R = 2cal/molk)
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers