Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
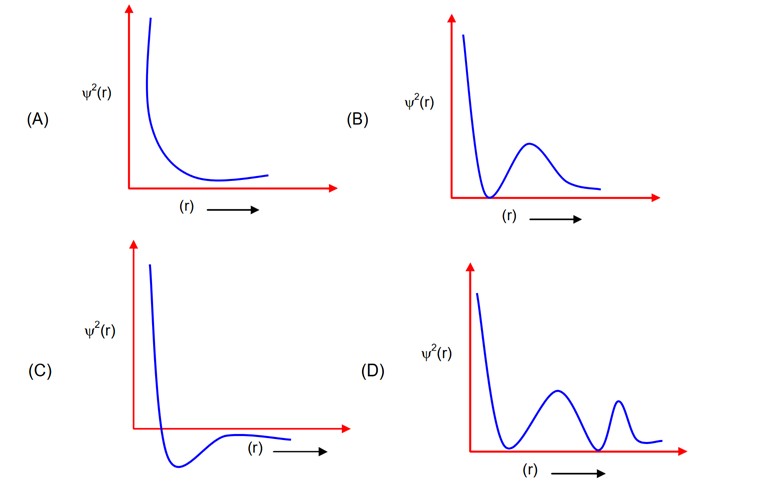
For 2S, number of radial moles = and will always be positive
it has one radial node and it is positive.
New answer posted
8 months agoContributor-Level 10
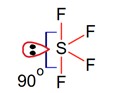
Geometry of SF4 is trigonal bipyramidal, in which there is one lonepair which occupy equatorial position as,
There are two lone pair – bond pair repulsions at 90°
New answer posted
8 months agoContributor-Level 10
Values of principal quantum number, n = 1, 2, 3, .
Values of azimuthal quantum number,
Number of values of magnetic quantum number are
Values of spin quantum number are
Number of orbitals for particular value of
New answer posted
8 months agoContributor-Level 10
Empirical formula is C5H7N
Empirical mass = 81
Molecular mass = 162
So, molecular formula is C10H14N2
New answer posted
8 months agoContributor-Level 10
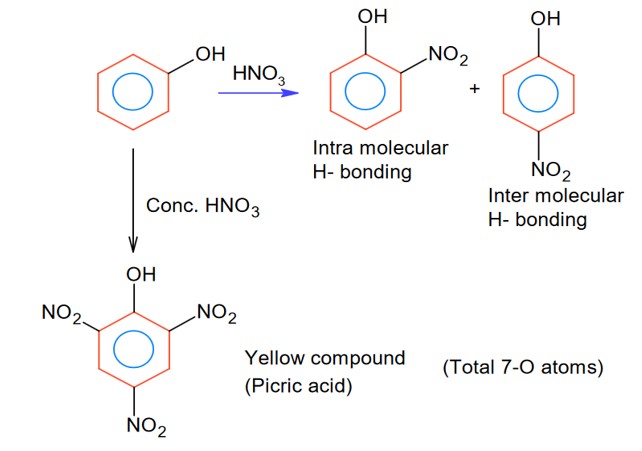
Intermolecular H- bonding and intra-molecular H- bonding producing compound may be the phenol derivatives.
New answer posted
8 months agoContributor-Level 10
Complete combustion of compound produces 0.2 gm CO2
Hence wt of carbon in 0.2 gm CO2
Therefore % of carbon in compound
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers