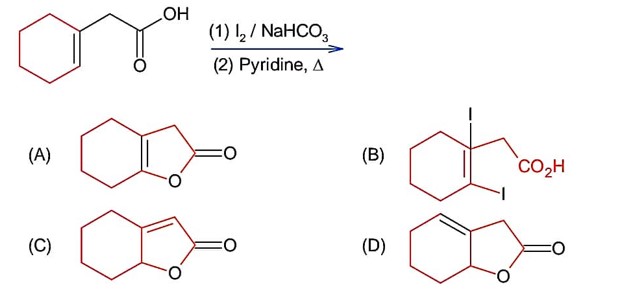
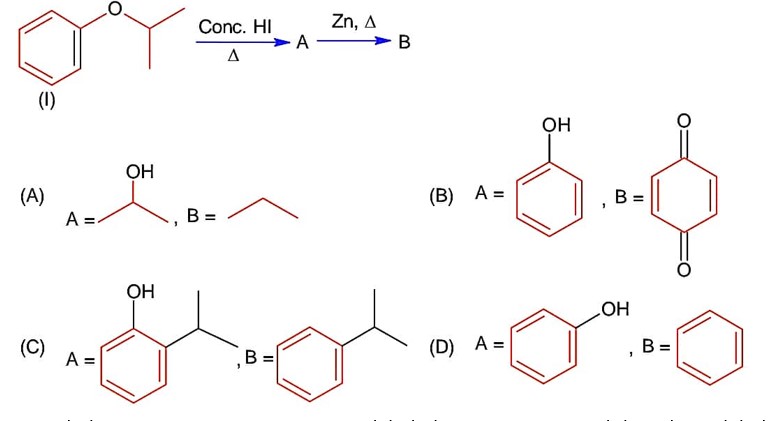
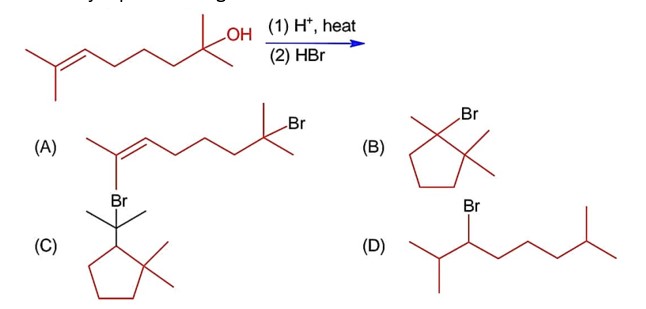
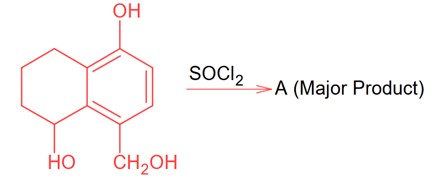
Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoNew answer posted
8 months agoContributor-Level 10
Tritium is radioactive and it decays into He3 during emission of β-radiation
1T3 → 2He3 + -1e0
New answer posted
8 months agoContributor-Level 10
Since SOCl2 is used to covert aliphatic (R-OH) into chlorides. It will not react with aromatic alcohol
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers