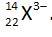Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
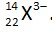
Number of electrons = 25
% of extra electrons =
New answer posted
8 months agoContributor-Level 10
It is reduction process, while rest are examples of disproportionation reaction.
New answer posted
8 months agoContributor-Level 10
PV = nmixRT
Let moles of He is x
Moles of H2 = 3 – x
4x + 2 (3 – x) = 10
x = 2 mol
Mass of He = 8 gm
New answer posted
8 months agoContributor-Level 10
Phenolphtalein is a pH dependant indicator. It is a weak acid which is colourless in acidic medium but gives pink colour in basic medium. The pink colour is due to its conjugate form. Phenolphthalein dissociates in basic medium. Therefore, assertion is true but reason is false.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers