Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
11.67
The acidic nature of phenol can be represented by the following two reactions:-
(a) Phenols react with sodium to give sodium phenoxide, liberating H2.
(b) Phenols react with sodium hydroxide to give sodium phenoxide and water as a by-product.
The acidity of phenol is more than that of ethanol. This is because phenol after losing a proton becomes phenoxide ion which undergoes resonance and is stabilized whereas ethoxide ion does not.
The resonating structures of phenoxide ion are shown as below:
The lone pair of electrons on oxygen delocalizes into the benzene (mesomeric effect) which reduces the electron density in the O-H bond. The
New answer posted
11 months agoContributor-Level 10
9.18. Auto-protolysis means self-ionisation of water. It may be represented as
2H2O(l) + H2O(l) ? H3O+(aq) + OH-(aq)
Acid 1 Base 2 Acid 2 Base 1
Due to auto-protolysis nature of water, it can act as an acid as well as base, i.e. amphoteric in nature.
New answer posted
11 months agoContributor-Level 10
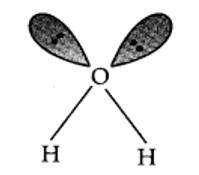
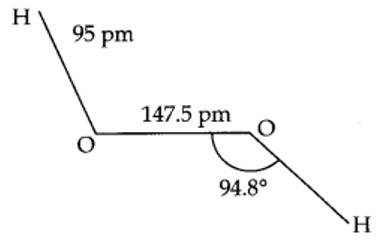
9.17. In water, O is sp3 hybridized. Due to stronger lone pair-lone pair repulsions than bond pair-bond pair repulsions, the HOH bond angle decreases from 109.5° to 104.5°. Thus, water molecule has a bent structure.
New answer posted
11 months agoContributor-Level 10
11.66
1. 1-phenylethanol from a suitable - The addition of water takes place according to Markovnikov rule. The alkene taken is styrene. And According to the rule, the positive charge i.e. H+ goes to the carbon of the double bond which has more number of hydrogens and the negative part i.e. OH- goes to the carbon that has less number of hydrogens. Therefore resulting the final product as 1-phenyl ethanol.
2. In the above conversion, NaOH gets dissociated into Na+ and OH-and Na+ then combines with Cl of chloromethylcyclohexane forming NaCl and thus the final product Cyclohexylmethanol is obtained.
3. In this conversion als
New answer posted
11 months agoContributor-Level 10
9.16. (i) BeH2< TiH2 < CaH2
(ii) LiH (iii) F—F < HH < DD (iv) H2O < MgH2
New answer posted
11 months agoContributor-Level 10
11.65
The reaction given below is:
Benzene reacts with concern. H2SO4 and undergoes the following mechanism:-
Step 1: The equilibrium produces SO3 in concentrated H2SO4, as shown below:
Step 2: SO3 is the electrophile which reacts with benzene to form arenium ion, as shown below:
Step 3: A proton is removed from the arenium ion to form benzenesulfonate ion.
Step 4: The benzenesulfonate ion accepts a proton to become benzene-sulphonic acid, as shown below:
Step 5: The benzene sulphonic acid then reacts with NaOH to give phenol as the final product, as shown below:
New answer posted
11 months agoContributor-Level 10
11.64
Step 1:- Protonation of ethene to form carbocation by electrophilic attack of H3O+.
Step 2:- Nucleophilic attack of water on carbocation.
Step 3:- Deprotonation to form ethanol.
New answer posted
11 months agoContributor-Level 10
There are many ways to this conversion. Two of them are given below:-
11.63
(a) In the above conversion, the chlorobenzene is treated with a base such as NaOH, KOH etc. (strong base). The base abstracts the hydrogen from the C-2 position (it can also abstract the hydrogen from the C-6 position, as both are equally acidic) leaving the negative charge at that position.
In the next step Cl- leaves, leaving behind the positive charge at that carbon. Both the negative charge and positive charge forms a bond resulting Benzyne as the intermediate.
After the formation of the Benzyne intermediate OH- of the base attacks at the C-1 position and fur
New answer posted
11 months agoContributor-Level 10
9.15. No. Because if saline hydrides react with water the reaction will be highly exothermic thus the hydrogen evolved in this case can catch fire. CO2 cannot be used as fire extinguisher because CO2 will get absorbed in alkali metal hydroxides.
New answer posted
11 months agoContributor-Level 10
9.14. HF is expected to have the highest magnitude of hydrogen bonding since, fluorine is most electronegative. Therefore, HF is the most polar.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers
