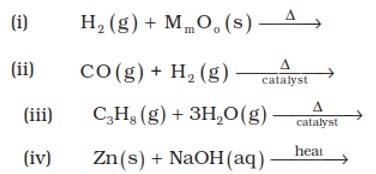Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
9.9. It is expected to be a Lewis acid. They are likely to accept electrons to become stable. They can form coordinate bond with electron rich compound.
2NaH(s) + B2H6 (g) → 2Na+[BH4]- (s)
&n
New answer posted
11 months agoContributor-Level 10
11.55
The structures of all isomeric alcohols of C5H12O are given below:
Naming is done by the conventional method. The -OH group is attached on the first carbon.
(b) 3-Methylbutan-1-ol
Butane is the longest chain and methyl is the substituent group.
(c) 3-Methylbutan-1-ol
the longest chain is butane and conventional naming method is used.
(d) 2,2-Dimethylpropan-1-ol
Isomer is made by transforming the principal carbon into tertiary type. The longest chain is butane and named accordingly.
(e) Pentan-2-ol
the longest chain is pentane and the numbering is chosen from the minimum position.
(f) 3-Methylbutan-2-ol
Butane is the longest chain,
New answer posted
11 months agoContributor-Level 10
9.8. (i) Electron deficient hydrides: Compounds in which central atom has incomplete octet, are called electron deficient hydrides. For example, BeH2, BH3 are electron deficient hydrides.
(ii) Electron precise hydrides: Those compounds in which exact number of electrons are present in central atom or the central atom contains complete octet are called precise hydrides e.g., CH4, SiH4, GeH4 etc. are precise hydrides.
(iii) Electron rich hydrides: Those compounds in which central atom has one or more lone pair of excess electrons are called electron rich hydrides, e.g., NH3, H2O.
New answer posted
11 months agoContributor-Level 10
9.7. This is due to its small atomic size and small bond length (74 pm) of H-H bond.H? H bond has very high bond enthalpy (435.9 kJ/mol) which results in low reactivity at room temperature. The reactivity is increased at high temperature or in presence of catalyst. Under these conditions, hydrogen reacts with many metals and non-metals to form hydrides.
New answer posted
11 months agoContributor-Level 10
9.6. (i) 3H2?(g)+2MoO3? ? Mo2?O3?+3H2?O(l)
(ii) CO (g) + H2 (g) ? CH3OH
(iii) C3H8 (g) + 3H2O(g) ? 3CO + 7H2(g)
(iv) Zn (s) + NaOH (aq) ? Na2ZnO2(s) + H2(g)
New answer posted
11 months agoContributor-Level 10
11.54
Butane is the longest chain.
Propane is principle chain and substituents are numbered accordingly.
Hexane is the longest chain. There are three -OH substituents and 2 methyl groups.
-OH of phenol is numbered a 1.
Propane is the principle chain and the alkoxy group is ethyl group.
the longest chain is pentane and substituents are numbered accordingly.
In such cases, cyclo groups are named first, followed by conventional naming methods.
The cyclo group is named first, followed by conventional naming methods.
The longest chain is butane when looked for minimum numbering case.
Cyclo group is named first. Pentane having a double bond is named
New answer posted
11 months agoContributor-Level 10
11.54
1. 2,2,4-Trimethylpentan-3-ol
The naming of the compound usually starts with numbering the carbons in the chain. The lower set of locants are chosen for this purpose, while in this case numbering under this condition is done from the left side. Once the carbons are mentioned, the position of -OH group is numbered and -ol is added as the suffix.
2. 5-Ethylheptane-2,4-diol
Here the longest chain is the straight chain. For such situations, numbering should be such that the functional groups should be denoted by the smallest number. Ethyl group is named at the beginning as it's a side chain.
3. &n
New answer posted
11 months agoContributor-Level 10
9.5. In bulk, dihydrogen can be produced by electrolysis of acidified water using Platinum electrodes.
2H2O (l) → H2 (g) + O2 (g)
Electrolyte is added to increase the dissociation of water.
New answer posted
11 months agoContributor-Level 10
9.4. The production of dihydrogen in coal gasification can be increased by reacting CO present in syngas mixtures with steam in the presence of iron chromate as the catalyst. This is called the water-gas shift reaction. Synthesis gas or 'syngas' is produced from sewage, sawdust, scrap wood, newspapers etc. The process of producing 'syngas' from coal is called 'coal gasification'.
&nb
New answer posted
11 months agoContributor-Level 10
9.3. In diatomic form, the K-shell of hydrogen is complete (1s2) and so it is quite stable. That is why hydrogen occurs in a diatomic form rather than in a monoatomic form.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers