Chemistry
Get insights from 6.9k questions on Chemistry, answered by students, alumni, and experts. You may also ask and answer any question you like about Chemistry
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
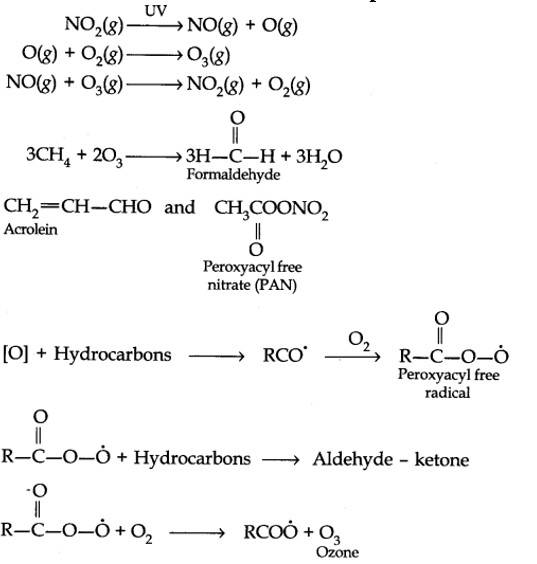
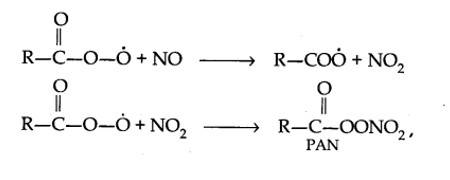
Harmful effects of photochemical smog: Photochemical smog causes serious health problems. Both ozone and PAN (peroxyacetyl nitrate) act as powerful eye irritants. Ozone and nitric oxide irritate the nose and throat and their high concentration causes headache, chest pain, dryness of the throat, cough and difficulty in breathing. Photochemical smog leads to cracking of rubber and extensive damage to plant life. It also causes corrosion of metals, stones, building materials, rubber and painted surfaces.
Control:
- Control of primary precursors of photochemical smog, such as NO2 and hydrocarbons, the secondary precursors such as ozone and PAN
New answer posted
11 months agoContributor-Level 10
3.27. (a) Element belonging to nitrogen family (group 15) e.g., nitrogen.
(b) Element belonging to alkaline earth family (group 2) e.g., magnesium.
(c) Element belonging to oxygen family (group 16) e.g., oxygen.
New answer posted
11 months agoContributor-Level 10
The word smog is a combination of smoke and fog. It is a type of air pollution that occurs in many cities throughout the world. Classical smog occurs in a cool, humid climate. It is also called reducing smog. Whereas photochemical smog (photo means light) occurs in warm and dry sunny climates. It has a high concentration of oxidising agents and therefore, it is also called oxidising smog.
New answer posted
11 months agoContributor-Level 10
1.24 Ferromagnetic substances would make better permanent magnets because when the ferromagnetic substance is placed in a magnetic field, all domains get oriented in the direction of magnetic field and strong a magnetic effect is produced.
New answer posted
11 months agoContributor-Level 10
This is mainly due to the large number of industries and power plants in the nearby areas. Acid rain has vapours of sulphuric acid dissolved in it. When it comes in contact with various statues or monuments, the acid reacts chemically with marble (calcium carbonate).
CaCO3 + H2SO4 à CaSO4 + H2O + CO2
As a result, the monument is being slowly disfigured, and the marble is getting discoloured and lustreless.
New answer posted
11 months agoContributor-Level 10
3.26.
Metals | Non-metals |
1. They have strong tendency to lose electrons to form cations. | 1. They have strong tendency to accept electrons to form anions. |
2. Metals are strong reducing agents. | 2. They are strong oxidising agents. |
3. Metals have low ionization enthalpies. | 3. Non-metals have high ionization enthalpies. |
4. They form basic oxides and ionic compounds. | 4. They form acidic oxides and covalent compounds. |
New answer posted
11 months agoContributor-Level 10
CO2 is mainly responsible for the greenhouse effect. Other greenhouse gases are methane, nitrous oxide, water vapours, CFCs (chlorofluorocarbons) and Ozone.
New answer posted
11 months agoContributor-Level 10
Carbon monoxide binds to haemoglobin to form carboxyhaemoglobin, which is about 300 times more stable than the oxygen-haemoglobin complex. In blood, when the concentration of carboxyhaemoglobin reaches about 3–4 per cent, the oxygen-carrying capacity of blood is greatly reduced. This oxygen deficiency results into headache, weak eyesight, nervousness and cardiovascular disorder. This is the reason why people are advised not to smoke. In pregnant women who have the habit of smoking the increased CO level in blood may induce premature birth, spontaneous abortions and deformed babies. On the other hand, CO2 does not combine with ha
New answer posted
11 months agoContributor-Level 10
1.23 An n-type semiconductor conducts because of the presence of extra electrons. Therefore, a group 14 element can be converted to n-type semiconductor by doping it with a group 15 element.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers