Class 11th
Get insights from 8k questions on Class 11th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 11th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
8 months agoContributor-Level 10
Here, total meq of acetic acid = 50 * 0.1 = 5
And total meq of NaOH = 25 * 0.1 = 2.5
After neutralization process
Meq of left acetic acid = 2.5
And meq of formed CH3COONa = 2.5
New answer posted
8 months agoContributor-Level 10
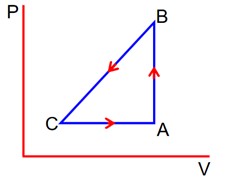
Given
QAB = +40J
QBC = 0J
QCA = -60J
WCA =?
WBC = -50J (on the gas)
UA = 1560J
1st Law on path A to B
WAB +
Þ 0 +
Þ UB = UA + 40
= 1560 + 40
UB = 1600 J
1st Low on path B
New answer posted
8 months agoContributor-Level 10
0.5 % KCl solution has molality (m) =
1 - α α α
And I =
1.976 = 1 + α
% = 97.6%
the nearest 98.
New answer posted
8 months agoContributor-Level 10
2 * mmol of l2 = 0.4
mmol of l2 = 0.2 mmol
mmol of Cu2+ = 0.2 * 2 * 10-3
New question posted
8 months agoNew question posted
8 months agoTaking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers