Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
7 months agoContributor-Level 10
The magnetic moment of an ion is determined by its number of unpaired electrons (n). For the given lanthanide ions:
o Ce? has 0 unpaired electrons (4f? ).
o Eu³? has 6 unpaired electrons (4f? ).
o Gd³? has 7 unpaired electrons (4f? ).
Gd³? has the highest number of unpaired electrons and therefore the highest magnetic moment.
New answer posted
7 months agoContributor-Level 9
Solubility of CdSO? is water ; S = 8 * 10? M
Using Ksp = S²
Ksp = 64 * 10? M
Now,
CdSO? Cd²? + SO? ²?
in H? SO? (0.01M)
Ksp = [Cd²? ] [SO? ²? ]
64 * 10? = S? (S? + 0.01)
S? << 0.01
So, S? = 64 * 10? M.
New answer posted
7 months agoContributor-Level 9
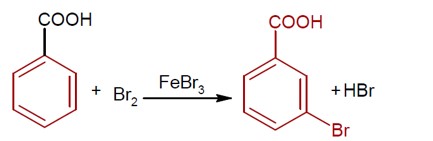
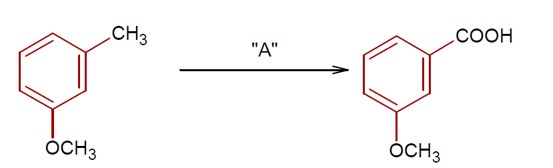
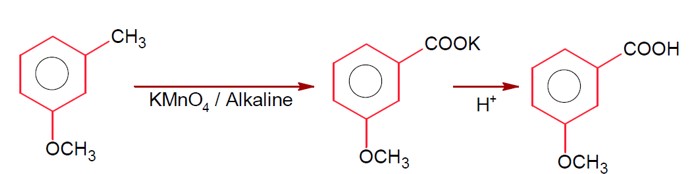
Moles of benzoic acid = 6.1 / 121 = 0.05
Theoretical moles of m- bromobenzoic acid = 0.05
Observed moles of m- bromobenzoic acid = 7.8 / 200 = 0.039
% yield = (0.039 / 0.05) * 100 = 78%
New answer posted
7 months agoContributor-Level 9
Half life, t? /? = 1min
Let, time of 99.9% completion of reaction be 't' min
Let the reaction is of first order
K = (2.303/t) log? ( [R]? / [R])
[R] = 0.001 [R]?
t = (2.303 * 3 min) / 0.693
t = 9.99 min
the nearest integer is 10.
New answer posted
7 months agoContributor-Level 9
λ° (BaCl? ) = 280 Scm²mol? ¹
λ° (H? SO? ) = 860 Scm²mol? ¹
λ° (HCl) = 426 Scm²mol? ¹
λ° (BaSO? ) = λ° (BaCl? ) + λ° (H? SO? ) - 2λ° (HCl)
= 280 + 860 - 2 * 426
= 288 Scm² mol? ¹
New answer posted
7 months agoContributor-Level 9
In Tollen's test for aldehyde, aldehyde is oxidized to carboxylic acid salt as:
R – CHO + H? O →R – COO? + 3H? + 2e?
So; 2e? are transferred per aldhyde group.
New answer posted
7 months agoContributor-Level 9
Partial hydrolysis of XeF? gives XeO? F?
XeF? + 2H? O → XeO? F? + 4HF
Compound A is XeF? , so number of lone pair on Xe is 1.
New answer posted
7 months agoContributor-Level 10
The reduction of aluminum oxide (Al? O? ) is performed through an electrolytic process in its molten state. This method is necessary because Al? O? is a highly ionic and stable compound.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 687k Reviews
- 1800k Answers