Class 12th
Get insights from 11.9k questions on Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
10 months agoContributor-Level 10
33. Option (ii) and (iv)
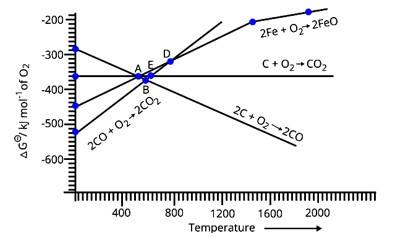
From Fig.6.1, at point B, all three lines, i.e. for the lines for all three given reactions, are below that of the line of reduction of FeO. This means, that the value of ΔG for the equivalent temperature at point B for reduction of FeO is greater than (positive) that of values of ΔG for all three reactions (negative). Thus, FeO will get reduced by all three reactions at point B. Same is the case with point E as that of point B, the value of ΔG for reduction of FeO at E is greater than the value of ΔG&nb
New answer posted
10 months agoContributor-Level 10
11. Cr (24)= [Ar]3d54s2
Zn (30) = [Ar] 3d104s2
The stability of orbitals according to the electrons filled is in order:
Fully filled > half-filled > partially filled
As 3d10 is completely filled orbital of Zn is more stable than the 3d5 half-filled orbital of Cr . It is comparatively easy to remove electron from 3d5 than from 3d10.
Therefore, the first ionization enthalpy of Cr is lower than that of Zn.
New answer posted
10 months agoContributor-Level 10
32. Option (i)
According to the above graph, at point D the equivalent value of ΔG for the reduction of FeO is approximately 330 units for the particular temperature. Also, for CO the equivalent value of Δ? at the particular temperature for point D is around −330 units. Thus, if we calculate the overall value of ΔG for the reduction of FeO with carbon monoxide then it is almost zero at point D.
New answer posted
10 months agoContributor-Level 10
31. Option (i)
Below point A, only the value of ΔG (CO, CO2) is less than the value of ΔG (Fe, FeO) at the corresponding temperatures. Thus, only carbon monoxide will be able to reduce FeO to Fe and will get itself oxidized into CO2.
New question posted
10 months agoNew answer posted
10 months agoContributor-Level 10
29. Option (i)
The cyanide process involves 3 steps:
First step - The finely grounded ore of gold and silver are made to come in contact with the solution containing the cyanide,
Second step - it involves separation of gold and silver from the cyanide solution
Third step - it involves the recovery of gold and silver in their pure forms precipitating the remaining solution with zinc dust.
Thus, the metal is recovered by displacing Zn with the metal (Au or Ag) from metal ions.
New answer posted
10 months agoContributor-Level 10
28. Option (i)
The electrolytic method can be used to purify zinc and copper. The impure metal is used as an anode in this method. As the cathode, a pure strip of the same metal is used. They are immersed in an appropriate electrolytic bath containing a soluble salt of the same metal.
The more basic metals remain in the solution, while the less basic metals are transferred to the anode mud.
New answer posted
10 months agoContributor-Level 10
27. Option (ii)
Reaction involved in the metallurgy of aluminum is 2Al2O3 + 3C→ 4Al + 3CO2.
The reaction at cathode is Al3+ + 3e−→ Al
The reaction at anode is
C+12O2 →CO + 2e and C+O2→CO2 + 4e−
Hence, from the reaction graphite anode is oxidized to carbon monoxide and carbon dioxide.
New answer posted
10 months agoContributor-Level 10
10. Mn has half-filled 3d5 electrons and Zn has fully-filled 3d10 electrons which give them extra stability and they both resist to lose electrons and get reduced.
The E? value also depends on the hydration enthalpy. More negative is the enthalpy of hydration, high is the E? value. This is the reason behind Mn, Ni and Zn are having higher E? value than expected
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 688k Reviews
- 1850k Answers