Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: C
The chelate effect is when a chelating ligand coordinates the stabilisation of a molecule. Any ligand that binds to metal forms a ring with 5 or 6 members, which is the most stable.
Here, CO, CN, H2O is monodentate ligand [Fe (C2O4)3]3- is an oxalate ligand.
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: A
Diamminedichloridoplatinum (II) is also known as azane
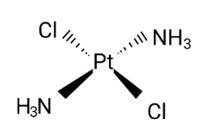
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: D
When 1 mol [CrCl3 (H2O)3]6H2O is treated with excess of AgNO3, 3 mol of AgCl is produced, i.e., [CrCl3 (H2O)3]6H2Ois dissociated in aqueous solution and all three chloride comes in solution.
[Cr (H2O)6]Cl3→ [Cr (H2O)6]3+ + 3Cl-
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: B
When 0.1 mol CoCl3 (NH3)5 is treated with excess of AgNO3, In the given reaction [CoCl3 (NH3)5]Cl2
then electrolytic solution must contain
[CoCl3 (NH3)5]Cl2+ and two Cl- ions.
Hence, it is 1:2 electrolyte.
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct option: A
Strong field ligands have five degenerate energy levels, which means they have more energy separation than weak field ligands.
Here, ΔE=
ΔE α
α
The wavelength decreases as the energy separation rises.
New answer posted
11 months agoContributor-Level 10
This is a Multiple Choice Questions as classified in NCERT Exemplar
Ans: Correct options: B
The bigger the value of constant, the better will be the stability.
Here, has the highest value of logk which corresponds to the highest value of k.
New answer posted
11 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Ambidentate ligands are ligands which have two donating sites. Coordinating compounds containing ambidentate ligands show linkage isomerism due to two different binding positions. Linkage isomerism have same ligand and geometry attached to a central metal ion by different donating sites
Examples:
(i)
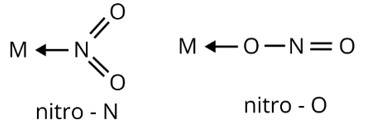
(ii) M←SCN
thiocyanato
M←NCS
isothiocyanato
New answer posted
11 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: In CuSO4⋅5H2O, water acts as ligand and causes crystal field splitting. This makes d - d transitions possible. On the other hand, in CuSO4, splitting is not possible due to the lack of ligand in the crystal field. As a result, no colour is visible.
New answer posted
11 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: Due to the presence of weak and strong field ligands in complexes, compounds with comparable geometry have distinct magnetic moments. The magnetic moment decreases when the CFSE increases, and vice versa.
Example: [Fe (CN)6]3- - Fe3+ ,3d5, CN- (strong field ligand, pairs electron)
[Fe (H2O)6]3- - Fe3+ ,3d5, H2O (weak field ligand, does not pair)
New answer posted
11 months agoContributor-Level 10
This is a Short Answer Type Questions as classified in NCERT Exemplar
Ans: CFSE is higher when the complex contains strong field ligand. Thus, crystal field splitting energy increases in the order
[Cr (Cl)6]3−< [Cr (NH3)6]3+< [Cr (CN)6]3−.
Because according to spectrochemical series the order of field strength is
Cl−
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers
