Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
1- When 3-methylaniline treated with NaNO2 + HCl it gets converted into chlorine complex.
When that complex reacted with HBF4 It gets converted into Barium Fluoride complex. This complex reacts with NaNO2 in presence of copper to give 3-Nitrotoluene.
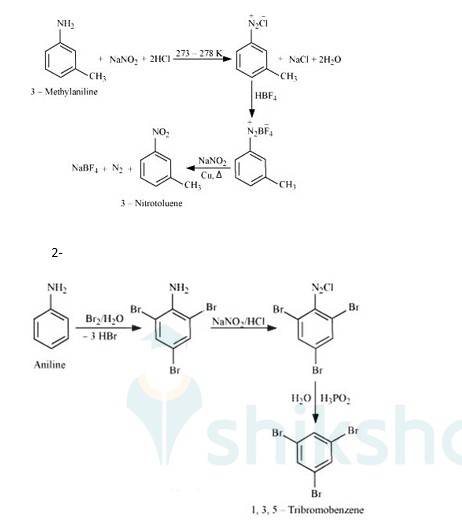
When aniline reacts with Br2 water it gets converted into 2,4,6 tribromobenzamine. When this further reacted with NaNO2/HCl it forms Chloride complex. This complex forms 1,3,5 tribromobenzene after treating with H3PO2 in presence of water.
New answer posted
a year agoContributor-Level 10
8.12 Electronic configuration of Mn2+ is [Ar]183d5 and Electronic configuration of Fe2+ is [Ar]18 3d6 . It is known that half-filled and fully-filled orbitals are more stable. Therefore, Mn in (+2) state has a half-filled stable configuration, whereas the Fe in +3 oxidation state has partially filled subshells, which are relatively unstable. This is the reason Mn2+ shows resistance to oxidation to Mn3+. Also, Fe2+ has 3d6 configuration and by losing one electron, it attains half- filled stable Hence, Mn2+ compounds more stable than Fe2+ towards oxidation to their +3 state.
Mn+2→
Manganese has the atomic no. 25 and its electronic config
New answer posted
a year agoContributor-Level 10
The different isomers of the molecular formula: C3H9N are given in the table. However only 1° amines will liberate nitrogen gas on the treatment with h=the nitrous acid are given as follows:
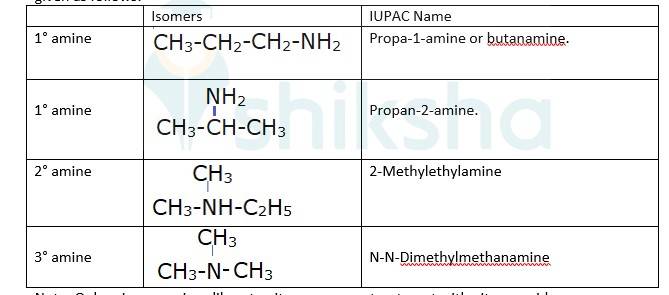
Note: Only primary amines liberate nitrogen gas on treatment with nitrous acid.
New answer posted
a year agoContributor-Level 10
5.33
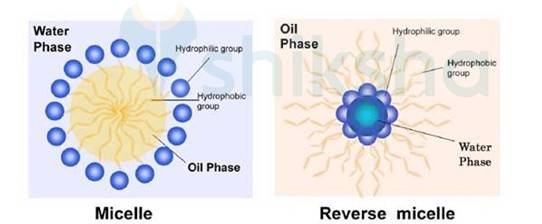
Soaps contain hydrophobic and hydrophilic part when dissolved in the water they arrange themselves in such a way that they form a spherical structure having hydrophobic part towards the centre and hydrophilic part away from centre. This cluster is known as Micelle. Ex-Sodium stearate + Water
(CH3 (CH2)16COO-Na + H2O)
New answer posted
a year agoContributor-Level 10
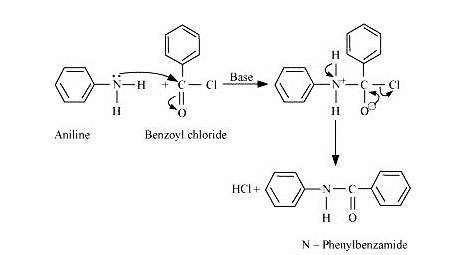
When aniline is treated with benzoyl chloride in the presence of base it gets converted into N-Phenylbenzamide.
New answer posted
a year agoContributor-Level 10
5.32
Uses of Emulsions-
It is used in making of medicines,
Cleansing action of soaps is based on this emulsion
Digestion of fats in intestine takes place by the process of
Antiseptics and disinfectant added to water form emulsion for
New question posted
a year agoNew answer posted
a year agoContributor-Level 10
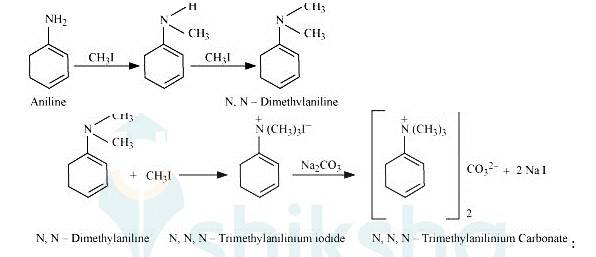
On excessive alkylation with methyl iodide aniline gets converted into N, N-Trimethylanilinium iodide. After reacting it with sodium carbonate it get converted into N, N-Trimethylanilinium carbonate.
New answer posted
a year agoContributor-Level 10
8.11 (i) The atomic number of Cr is 24 and the electronic configuration is [Ar] 3d54s1. When 3 electrons are removed, it becomes Cr3+. The electronic configuration of Cr3+: 1s2 2s2 2p6 3s2 3p6 3d3 Or [Ar]3d3
(ii) The atomic number of Pm is 61 and the electronic configuration is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 4f5 5s2 5p6 6s2 or [Xe] 4f56s2. When 3 electrons are removed, it becomes Pm+3, having the electronic configuration,
Pm3+: 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 4f4
Or, [Xe]544f4
(iii) The atomic number of Cu is 29 and has the electronic configuration of [Ar] 3d104s1. On removing one electron, the Cu+
New answer posted
a year agoContributor-Level 10
5.31
Electrophoresis: The movement of colloidal particles under an applied electric potential is called When an electric potential is applied to two platinum electrodes dipping in colloidal solutions the colloidal particle move towards the Oppositely charged electrodes.
Coagulation: The process of settling of colloidal particles is called coagulation. When the charge is removed from colloidal solution somehow, then particle start coagulation and settling due to the force of
Dialysis: It is a process of removing a dissolved substance from AC colloidal solution by means of diffusion through a suitable the animal membrane or parchment paper
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers
