Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
5.30
The catalytic reaction that depends upon the pore structures of the catalysts and the size of reactant and product molecules is called shape selective catalysis. Zeolites are good shape- selective catalysts.
New answer posted
a year agoContributor-Level 10
8.10 The 5f orbitals (in actinoids) have a poorer shielding effect than 4f orbitals (in lanthanoids). Thus, the effective nuclear charge experienced by electrons in outer shells in case of actinoids is much more that experienced by lanthanoids.
As the effective nuclear charge experienced is high, the electrons are attracted with much force, hence the size of the atom decreases. Hence, actinoid contraction is greater from element to element than lanthanoid contraction.
New answer posted
a year agoContributor-Level 10
5.29
Catalysis by zeolites is dependent on shape. Because zeolites are shape-selective catalysts. They are alumino silicates which are microporous in nature. It has Honeycomb structure. That makes them shape selective. In zeolites, some si atoms are replaced by Al to form Al-O-Si network.
Reactants are very sensitive to the pore size of zeolites. Zeolites are used in petrochemical industry. Ex- ZSM-5 used to convert alcohol into gasoline.
New answer posted
a year agoContributor-Level 10
(i) CH3CH2CH2NH2 + HCl → CH3CH2CH2N+H3Cl-
The final product is (N-propyl ammonium chloride.)
(ii) (C2H5)3N + HCl → (C2H5)3N+HCl-
The final product is (Tri ethyl ammonium chloride)
New answer posted
a year agoContributor-Level 10
5.28
Activity - It is the ability of the catalyst to accelerate the Reaction. It mostly depends upon the Chemisorption strength.
Selectivity - It is an ability to direct reaction to yield of a particular product i.e., One catalyst cannot be a catalyst for other reactions.
New answer posted
a year agoContributor-Level 10
8.9 The stability in aqueous condition depends on the hydration energy of the ions when they bond to the water molecules. And, the hydration energy is the amount of heat released as an ionic substance is dissolved and its constituent ions are hydrated or surrounded by water molecules.
Now, in Cu2+ and Cu+ ion, Cu2+ has a greater charge density than the Cu+ ion and so forms much stronger bonds releasing more energy. Therefore, in an aqueous medium, Cu2+ ion is more stable than Cu+ ion. This is because the energy required to remove one electron from Cu+ to Cu2+, is compensated by the high hydration energy of Cu2+.
New answer posted
a year agoContributor-Level 10
1- Alkyl group contribute inductive effect which increases the basic strength of
NH3
Then C6H5NH2 is having –I effect that reduces strength. And C6H5CH2NH2 increases the basic strength but not as much as C2H5 group.
Hence final order will be C6H5NH2<
2- By taking into consideration –R effect and steric hindrance of groups we can arrange them in the order
C6H5NH2< C2H5NH2< (C2H5)3N< (C2H5)2NH.
Because (C2H5)3N has a lot of steric hindrances that reduces the basic strength.
3- In C6H5NH2, N is directly attached to the ring that causes delocalization of electrons of the benzene ring. Whereas in case of C6H5CH2NH2 it is not directly connected to benze
New answer posted
a year agoContributor-Level 10
Benzene into aniline
When Benzene is treated with HNO3/H2SO4 it forms nitrobenzene. When Nitrobenzene reduced with Sn/HCL it forms Aniline because Sn/HCl is a reducing Mixture.
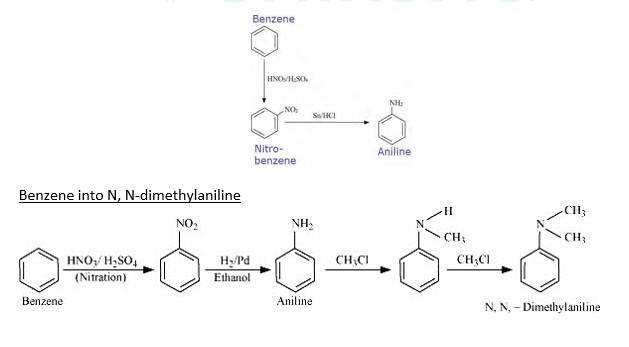
When Benzene is reacted with nitrating mixture it forms nitrobenzene. When it Reduced H2/Pd in ethanol or Sn/HCl, it forms Aniline. When Aniline reacts 2 times with CH3Cl It forms N, N- dimethylaniline.
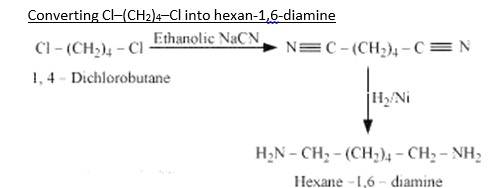
When 1,4-dichlorobutane reacts with NaCN it forms Di cyanide compound, After Hydrogenation it forms the Hexane 1,6-Diamine.
New answer posted
a year agoContributor-Level 10
5.27
Oxidation of sulphur dioxide into sulphur trioxide in the presence of
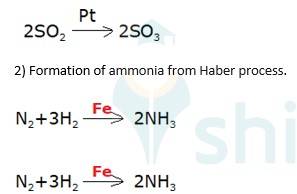
(3) Oxidation of ammonia into nitric acid in presence of platinum gauze in Ostwald's process
→ 4NH3 (g)+ 5O2 (g) Pt (s) 4NO +6H2O
(4) Hydrogenation of vegetable oils in presence of Nickel as catalyst
Vegetable oil (l)+H2 (g) Ni → Vegetable ghee (s)
New answer posted
a year agoContributor-Level 10
8.8 Given, Z=atomic number = 27 = [Ar]3d7 4s2
⇒ M2+ = [Ar]3d7
Hence, 3 unpaired electrons are present.
The spin only magnetic moment μ= √n(n+2)
Where n is the number of unpaired electrons.
Hence, μ =√(3*(3+2))
μ = √15 BM or μ = 3.87 BM
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers
