Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New question posted
a year agoNew answer posted
a year agoContributor-Level 10
8.16 (i) Vanadate (VO3 -)-Oxidation state of V is +
(ii) Chromate (CrO4 2-)-Oxidation state of Cr is +
(iii) Permanganate (MnO4 -) Oxidation state of Mn is + 7
New answer posted
a year agoContributor-Level 10
Pkb value is the negative logarithm of the basicity constant (Kb) .i.e., pKb = -logKb
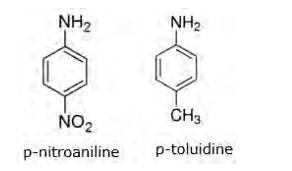
Evidently, smaller the value of pKb , stronger is the base (strong tendency to donate electrons). Aliphatic amines(R-NH2) are more basic(tendency to donate electrons) than aromatic amines(C6H5NH2) because of the following reasons:
=>In aliphatic amines, alkyl groups are present. Alkyl groups are electron releasing groups, hence they increase the elecron density of N-atom and thus is easily available to donate electrons. This poperty makes aliphatic amines more basic.
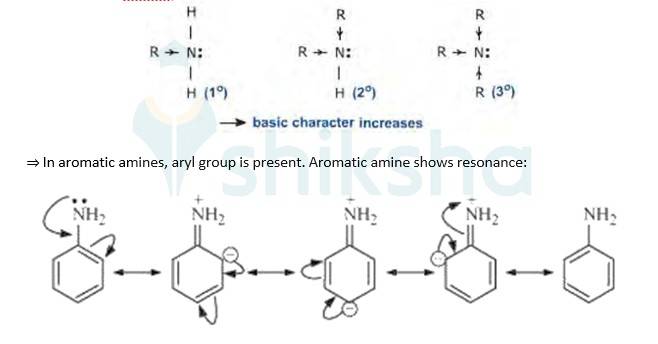
As a result of resonance, the lone pair of electrons on the nitrogen atom gets delocaliz
New answer posted
a year agoContributor-Level 10
7.16
Pt is a noble metal (inert) and does not react with oxygen, whereas Zn, Ti, and Fe react with quickly with oxygen.
New answer posted
a year agoContributor-Level 10
7.15
The oxygen atom has a smaller size and greater electronegativity than sulphur atom, hence intermolecular hydrogen bonding is possible in water (H2O). But, only weak Van there Waal's force holds H2S molecules, hence water is liquid and H2S is a gas.
New question posted
a year agoNew answer posted
a year agoContributor-Level 10
7.14
The enthalpy of dissociation of H-E bond (E= O, S, Se, Te, Po) in the hydrides decreases down the group. Hence, the thermal stability of the hydrides decreases down the group. The thermal stability is in the order H2O > H2S> H2Se> H2Te >H2Po.
New answer posted
a year agoContributor-Level 10
7.13
Sulphur generally exists in the combined form in the following sulphate minerals like gypsum (CaSO4.2H2O), Epsom salt (MgSO4.7H2O), barite (BaSO4) and sulphide minerals such as galena (PbS), zinc blende (ZnS), copper pyrites (CuFeS2).
Traces of sulphur are present in volcanoes as hydrogen sulphide. Sulphur is also present in organic materials such as eggs, proteins, garlic, onion, mustard, hair, and wool.
New answer posted
a year agoContributor-Level 10
7.12
H3PO3 on heating dissociates into orthophosphoricacid (H3PO4) and phosphine (PH3). Since, the oxidation states of orthophosphorous acid (H3PO3), orthophosphoric acid (H3PO4) and phosphine (PH3) are +3, +5 and -3 respectively, this reaction is known as a disproportionate reaction (since the oxidation states of one of the product are positive and the other is negative). The reaction is as follows.

New answer posted
a year agoContributor-Level 10
7.11
The basicity of a compound is defined as the number of acidic hydrogen atoms present in the compound. For a hydrogen atom to be acidic, it should be bonded to a strongly electronegative element like fluorine or oxygen atom.
As shown in the figure, 3 hydrogen atoms (green circled) are attached to an oxygen atom, which makes them acidic. Hence, the basicity of H3PO4 is 3.

Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers