Ncert Solutions Chemistry Class 12th
Get insights from 2.6k questions on Ncert Solutions Chemistry Class 12th, answered by students, alumni, and experts. You may also ask and answer any question you like about Ncert Solutions Chemistry Class 12th
Follow Ask QuestionQuestions
Discussions
Active Users
Followers
New answer posted
a year agoContributor-Level 10
7.1
Pentahalides (like MX5) means the compounds in which metal is bonded with five halogen atoms. Thus, the oxidation state of metal here is +5. Similarly, trihalides (like MX3) means the compounds in which metal is bonded with three halogen atoms.
Thus, the oxidation state of metal here is +3. Now, as the polarizing power is directly proportional to the charge, the metals with the higher charge will have higher polarizing power. Hence, Pentahalides are more covalent than trihalides.
Note: Polarizing power is the ability of a cation to distort an anion.
New answer posted
a year agoContributor-Level 10
8.15 For answering this question, we can compare the electronic configuration of standard elements and then write their corresponding oxidation
S. No | Electronic configurations in ground state | Stable oxidation states |
1 | 3d3 Vanadium | +2, +3, +4, +5 |
2 | 3d5 Chromium | +3, +4, +6 |
3 | 3d5 Manganese | +2, +4, +6, +7 |
4 | 3d8 Nickel | +1, +2, +3, +4 |
5 | 3d4 | 3d4 configuration is not stable at ground state |
New question posted
a year agoNew answer posted
a year agoContributor-Level 10
The best test for distinguishing methyl amine and dimethylamine is the Carbylamines test.
Carbylamine Test: Aliphatic and aromatic primary amines on heating with chloroform and ethanolic potassium hydroxide form foul-smelling isocyanides or carbylamines.
In this case, Methylamine (which is an aliphatic primary amine) gives a positive carbylamine test while dimethylamine wont.
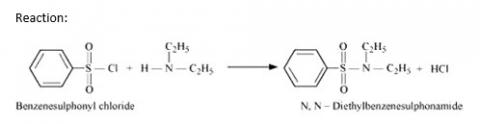
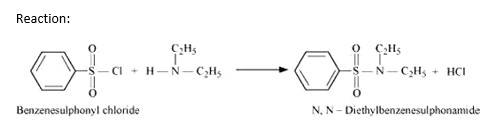
Hinsberg's reagent (benzenesulphonyl chloride, C6H5SO2Cl). can be used to distinguish secondary and tertiary amines.
Hinsberg Test: Secondary amines react with Hinsberg's reagent to form a product that is insoluble in an alkali. For example, N, N? diethylamine reacts
New answer posted
a year agoContributor-Level 10
8.14 The elements in the first-half of the transition series exhibit many oxidation states with Mn exhibiting a maximum number of oxidation states (+2 to +7). The stability of +2 oxidation state increases with the increase in atomic number. This happens as more electrons are getting filled in the d-orbital.
However, Sc ( [Ar] 3d14s2) does not show +2 oxidation state, instead, it loses all the three valence electrons to form Sc3+. The +3 oxidation state of Sc is very stable as it attains stable configuration.
For Mn ( [Ar] 3d54s2), +2 oxidation state is very stable because after losing two electrons, it attains stable half-filled str
New answer posted
a year agoContributor-Level 10
5.35
We can say that colloid is not a substance but a state of a substance which is dependent on the size of particle colloidal state is intermediate between a true solution and a suspension.
When a size of substance is between 1nm to1000nm it behaves as colloid otherwise not.
New answer posted
a year agoContributor-Level 10
1 - Methylethanamine
The root name is based on the longest chain with the -NH2 attached. The chain is numbered so as to give the amine unit the lowest possible number. The longest chain is ethane chain which is further suffixed with 'amine'.
2 - Propan-1-amine
The longest chain here is propane. The naming is such that amine unit should get a the lowest possible number. Propane-1-amine can also be written as 1-propylamine.
3 - N−Methyl-2-methylethanamine
The chain is numbered so as to give the amine unit the lowest possible number. The other alkyl group is treated as a substituent, with N as the locant. The N locant is listed before numeri
New answer posted
a year agoContributor-Level 10
8.13 The oxidation states displayed by the first half of the first row of transition metals are given in the table below.
METALS | Sc ( [Ar] 3d14s2) | Ti ( [Ar] 3d24s2) | V ( [Ar] 3d34s2 ) | Cr ( [Ar] 3d54s1) | Mn ( [Ar] 3d54s2) |
OXIDATION STATES |
| +2 | +2 | +2 | +2 |
+3 | +3 | +3 | +3 | +3 | |
| +4 | +4 | +4 | +4 | |
|
| +5 | +5 | +6 | |
|
|
| +6 | +7 |
Except for Sc, all others metals display +2 oxidation state. This is because as the atomic number increases, the number of electrons in the valence shell increases. +2 oxidation state is attained by the loss of the two 4s electrons by these metals. As the number of electron increases, the possibility of an ion with +2 oxidation state being stable (by attaining half-filled structure) also increases. Finally, Mn2+ ions have half-filled structure and are very stable.
New answer posted
a year agoContributor-Level 10
5.34
Alcohol- a colloidal solution having alcohol as the dispersion medium and a solid substance as the dispersed Ex- colloidal sol of cellulose nitrate in ethyl alcohol.
Aerosol- a colloidal solution having gas as the dispersion medium and a solid substance as the dispersed Ex-Smoke.
Hydrosol- a colloidal solution having Water as the dispersion medium and a solid substance as the dispersed Ex-Gold sol.
Taking an Exam? Selecting a College?
Get authentic answers from experts, students and alumni that you won't find anywhere else
Sign Up on ShikshaOn Shiksha, get access to
- 66k Colleges
- 1.2k Exams
- 691k Reviews
- 1850k Answers